Supramolecular Assemblies of Polymetallic Porphyrins and Porphyrazines for Molecular Electronics and Devices
Henrique E. Toma*, Koiti Araki, Marcos M. Toyama and Gregoire J. F. Demets
Instituto de Química, Universidade de São Paulo,
CEP 05513-970, São Paulo, SP, Brazil
This is an abstract
for a presentation given at the
Ninth
Foresight Conference on Molecular Nanotechnology.
There will be a link from here to the full article when it is
available on the web.
Porphyrin and porphyrazine systems containing bridging groups can be conveniently modified with ruthenium polypyridine or ruthenium cluster groups, in order to generate a unique class of supermolecular species exhibiting multiple redox and photoactive centers [1]. Such a combination has been successfully exploited in our laboratory for catalytical and electrocatalytical applications, as well as for photoinduced electron transfer and energy transfer processes, particularly for PDT applications [2]. An important property of the cationic supramolecular systems is their ability to form homogeneous molecular films, allowing the assembly of bilayers by dip-coating with suitable anionic sulphonated porphyrins and phtalocyanines [3]. According to our molecular mechanics calculations, this type of supramolecular assembly requires a specially designed molecular architecture in order to minimize steric hindrance and to allow efficient stacking and molecular recognition, as illustrated in Fig. 1. The assembly involves the orientation of the pi-conjugated rings, which are kept together by means of strong hydrophobic intermolecular interactions, as well as by the electrostatic interactions between the peripheral cationic and anionic residues.
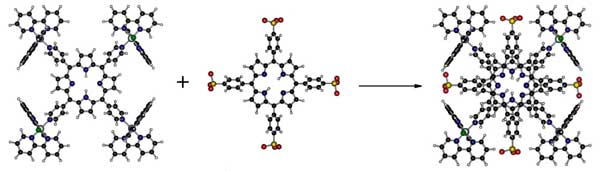
Fig. 1. Stacking of the tetraruthenium(bipyridine)porphyrin(4+) and tetrasulphonatedporphyrin(4-) species.
The organized films can be conveniently generated onto conducting surfaces, exhibiting rectifying response in the presence of redox active species, such as molecular oxygen, in addition to enhanced conductivity and photoaction response in the presence of light [3]. Interesting applications in electrochemical sensor devices [4], and as conducting molecular films have been developed in our laboratory. (Support: FAPESP and CNPq Agencies, Brazil)
References
- Toma, H. E. and Araki, K., Coord. Chem. Rev. 2000, 196, 307.
- Araki, K., Silva C.C., Toma, H. E., Catalani, L. H., Medeiros M. H. G., and Di-Mascio, P., J. Inorg. Biochem., 2000, 78, 269.
- Toyama, M. M., Demets, G. J. F., Araki, K. and Toma, H. E., Electrochem. Com., 2000, 2, 749
- Araki, K., Angnes L. and Toma H. E., Advanced Materials, 1995, 7, 554.
Abstract in RTF format 589,573 bytes
*Corresponding Address:
Henrique E. Toma
Instituto de Química, Universidade de São Paulo,
C.Postal 26077, Cep 05513-970, São Paulo, SP, Brazil
phone: 55 (11) 3818 3887
fax: 55 (11) 3815 5579
email: [email protected]
|