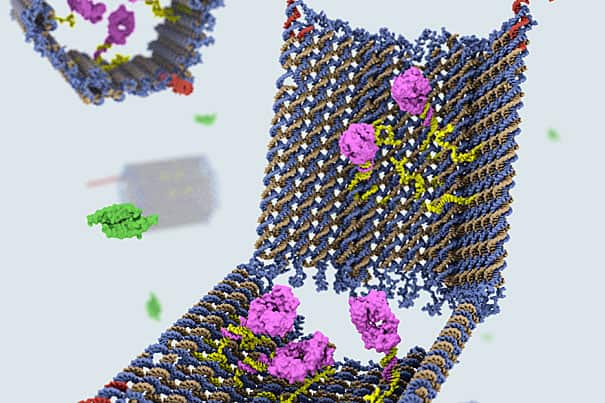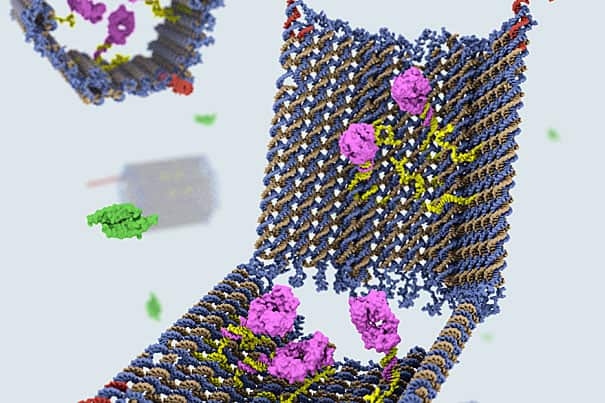
“The nanosized robot was created in the form of an open barrel whose two halves are connected by a hinge. The DNA barrel, which acts as a container, is held shut by special DNA latches that can recognize and seek out combinations of cell-surface proteins, including disease markers. This image was created by Campbell Strong, Shawn Douglas, and Gaël McGill using Molecular Maya and cadnano.“
DNA nanotechnology is not only a very promising path toward productive nanosystems and atomically precise manufacturing, but also a path to increasingly sophisticated DNA molecular machines for near-term drug delivery applications in nanomedicine. A recent advance comprises an autonomous DNA nanorobot incorporating a DNA origami chasis and DNA aptamer locks functioning as logical AND gates that are unlocked after the aptamers bind a protein target on the target cell, allowing the nanorobot to discharge its therapeutic cargo. A hat tip to KurzweilAI.net for reprinting this Harvard Gazette news release written by Twig Mowatt “Sending DNA robot to do the job: Technology has potential to seek out cancer cells, cause them to self-destruct“:
Researchers at the Wyss Institute for Biologically Inspired Engineering at Harvard University have developed a robotic device made from DNA that could potentially seek out specific cell targets within a complex mixture of cell types and deliver important molecular instructions, such as telling cancer cells to self-destruct. Inspired by the mechanics of the body’s own immune system, the technology might one day be used to program immune responses to treat various diseases. The research findings appear today in Science [“A Logic-Gated Nanorobot for Targeted Transport of Molecular Payloads” abstract; full text available for fair use on Church lab web site].
Using the DNA origami method, in which complex 3-D shapes and objects are constructed by folding strands of DNA, Shawn Douglas, a Wyss Technology Development Fellow, and Ido Bachelet, a former Wyss postdoctoral fellow who is now an assistant professor in the Faculty of Life Sciences and the Nano-Center at Bar-Ilan University in Israel, created a nanosized robot in the form of an open barrel whose two halves are connected by a hinge. The DNA barrel, which acts as a container, is held shut by special DNA latches that can recognize and seek out combinations of cell-surface proteins, including disease markers. When the latches find their targets, they reconfigure, causing the two halves of the barrel to swing open and expose its contents, or payload. The container can hold various types of payloads, including specific molecules with encoded instructions that can interact with specific cell surface signaling receptors.
Douglas and Bachelet used this system to deliver instructions, which were encoded in antibody fragments, to two different types of cancer cells — leukemia and lymphoma. In each case, the message to the cell was to activate its “suicide switch” — a standard feature that allows aging or abnormal cells to be eliminated. And because leukemia and lymphoma cells speak different languages, the messages were written in different antibody combinations. …
“We can finally integrate sensing and logical computing functions via complex, yet predictable, nanostructures — some of the first hybrids of structural DNA, antibodies, aptamers, and metal atomic clusters — aimed at useful, very specific targeting of human cancers and T-cells,” said George Church, a Wyss core faculty member and professor of genetics at Harvard Medical School, who is principal investigator on the project. …
A key feature of this work is that the DNA aptamer changes structure upon binding its target so it releases its hold on the complementary part of the DNA latch. Since two DNA latches hold the nanorobot in a closed configuration, the latches can be programmed to both respond to the same cell surface target, or to each respond to a different target so that both targets would need to be on the cell to activate the nanorobot to open and allow the payload molecules to bind their targets. This logical AND function allows for much greater specificity in recognizing target cells. As the authors conclude, “These findings demonstrate that the robots can induce a variety of tunable changes in cell behavior.” Conceivably a similar mechanism could be used in an atomically precise manufacturing operation in which DNA nanorobots could add a payload molecule to a workpiece depending on whether both of two specific molecular signals on the workpiece were present.
—James Lewis