A useful addition to the toolkits for nanomedicine and synthetic biology would be a cell membrane pore to exclude or admit predetermined molecules on demand. A hat tip to Kurzweil Accelerating Intelligence for describing and pointing to this UCL news release “DNA ‘building blocks’ pave the way for improved drug delivery“:
DNA has been used as a ‘molecular building block’ to construct synthetic bio-inspired pores which will improve the way drugs are delivered and help advance the field of synthetic biology, according to scientists from UCL and Nanion Technologies.
The study, published today in Nature Nanotechnology [abstract] and funded by the Biotechnology and Biological Sciences Research Council (BBSRC), Leverhulme Trust and UCL Chemistry, shows how DNA can be used to build stable and predictable pores that have a defined shape and charge to control which molecules can pass through the pore and when.
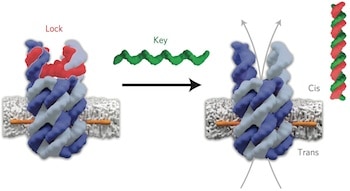
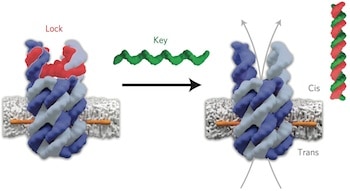
Lead author, Dr Stefan Howorka (UCL Chemistry), said: “Natural biological pores made of proteins are essential for transporting cargo into and out of biological cells but they are hard to design from scratch. DNA offers a whole new strategy for constructing highly specific synthetic pores that we can open and close on demand. We’ve engineered our pores to act like doors – the door unlocks only when provided with the right key. By building these pores into drug carriers, we think it will allow for much more precise targeting of therapeutics.”
Many therapeutics including anti-cancer drugs can be ferried around the body in tiny carriers called vesicles which are targeted to different tissues using biological markers. Previously, releasing the drugs from inside the vesicles was triggered with temperature-induced leaky vesicle walls or with inserted peptide channels, which are less rigid and predictable than DNA.
Using DNA building blocks, the team designed pores with pre-determined structures and defined properties which were precisely anchored into the walls – or membranes – of vesicles.
“Our pores take the shape of an open barrel made of six DNA staves. We designed a molecular gate to close off one entrance but then re-open the channel when a specific molecule binds. Anchors with high membrane affinity were attached to tether the water-soluble pores into the oily membrane,” explained first author, Dr Jonathan Burns (UCL Chemistry).
Using electrophysiology techniques, the researchers verified that the pore vertically spanned the surface of the membrane and was stable with an internal width of 2 nm, which is an appropriate size for small drugs molecules to fit through.
The gate’s lock and release mechanism was then tested with electrophysiology techniques as well as with fluorophores, which are of equivalent size to small molecules. As the DNA pore had a net negative charge, fluorophores with a net negative charge moved through with more ease than those with a net positive charge, showing selectivity for which cargo could exit. Removing the lock with a matching key increased of traffic 140-fold compared to a mismatched key.
Co-author Astrid Seifert who works with Dr Niels Fertig at Nanion Technologies, said: “We were able to precisely analyse the performance of each of the pores we created. We first inserted pores in membranes and then tested the biophysical response of each channel using advanced microchips. We’ve not only developed a new way to design highly specific pores but also an automated method to test their properties in situ, which will be important for testing pores being used for targeted drug delivery in the future.”
The researchers plan on testing the synthetic pores in a variety of scenarios including the release of anti-cancer drugs to cells and the development of pores that release pharmaceutically active biomolecules.
Dr Howorka added, “Our approach is a big step forward in building and using synthetic biological structures and promises a new era in pore design and synthetic biology. We have demonstrated such precise control over the behaviour of the pore, both in terms of selectivity and in terms of responsiveness that we believe that the method paves the way for a wide range of applications from drug delivery to nanosensing.”
A variety of applications are suggested by the ability to build such a precisely controlled cellular valve. It will be interesting to see which ones appear soon. Perhaps a point to clarify: how difficult will it be to insert such engineered pores into various types of cells in living animals/patients? How well do the pores hold up in the living organism?
—James Lewis, PhD