Summary
David Leigh starts with an investigation into the vision required for scientific progress. A scientist needs to find a problem interesting enough that is worth spending time on, yet sufficiently difficult that it hasn’t been solved. One must recognize that the time has come for solving that problem. Nanotechnology lost its way in the ‘90s due to over-complex ideas of nanofabrication.
He continues with a description of a molecular machine that can create stereoisomers in a programmable fashion. Finally, a nanoscale molecule assembler has become reality. Although it is simple, it is a new format of catalysis and represents a fundamentally different process. Leigh’s group is working on other molecular machines that take lessons from biology and improve upon them.
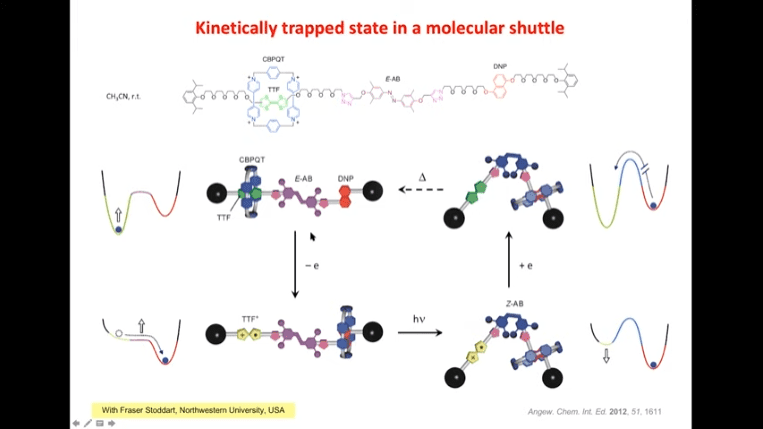
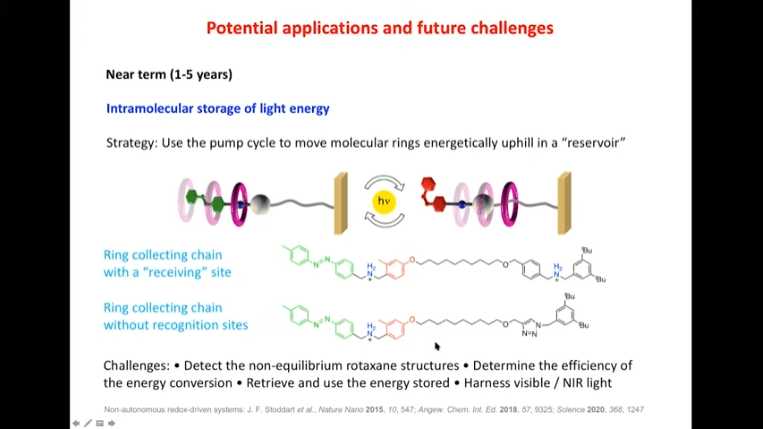
Next talk with Raymond Astumian was about thermodynamics vs. kinetics and a proposal for a mechanism that can catalyze chemical reactions using physical force. The mechanism is driven by redox reaction cycles and can occur at room temperature in a single vessel. It works by shifting the position of cyclophane rings during each redox phase to be closer to each other, physically forcing two molecules into close contact and bonding them together. This is very similar to how ATP synthase works.
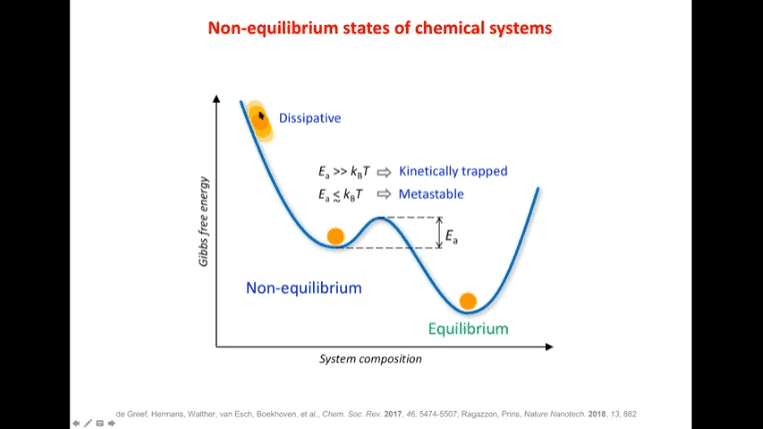
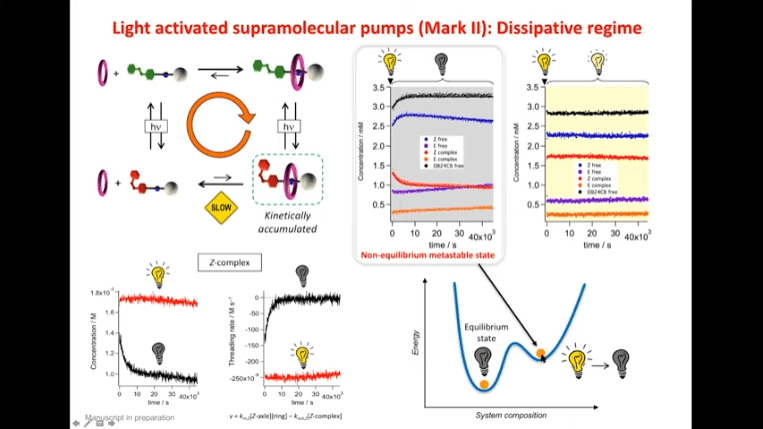
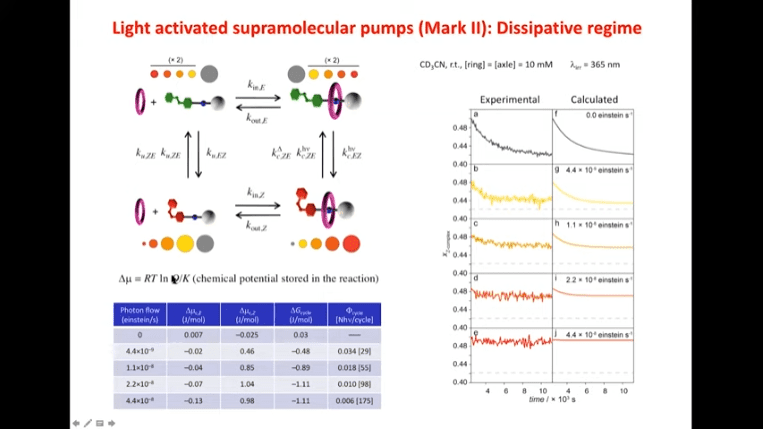
Alberto Credi – Most molecular devices exploit equilibrium properties – this is not surprising because the equilibrium state is the easiest state to obtain. However, living systems operate away from equilibrium. The systems that support life are non-equilibrium processes, and it’s typically not a good sign when an organism reaches equilibrium.
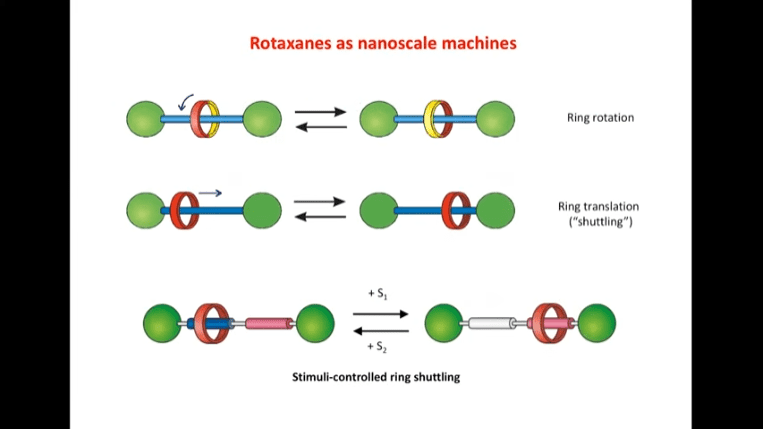
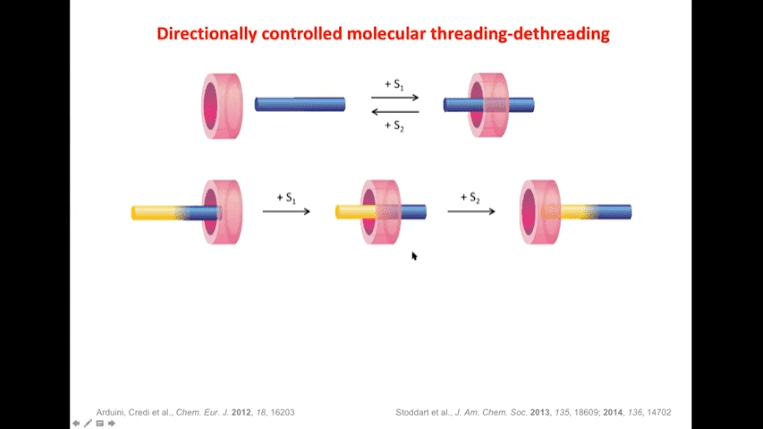
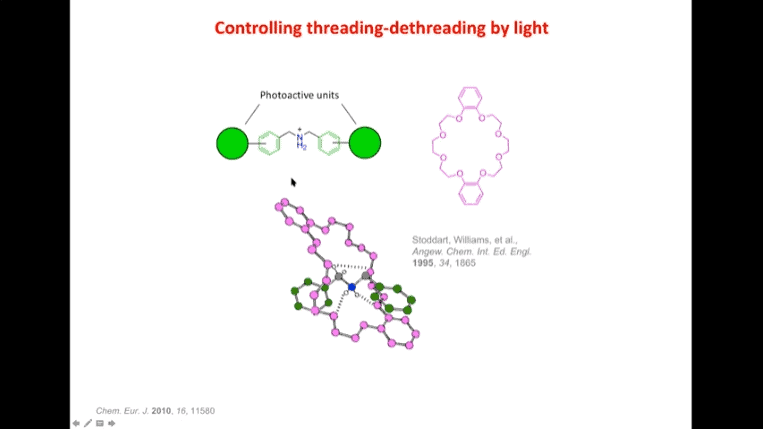
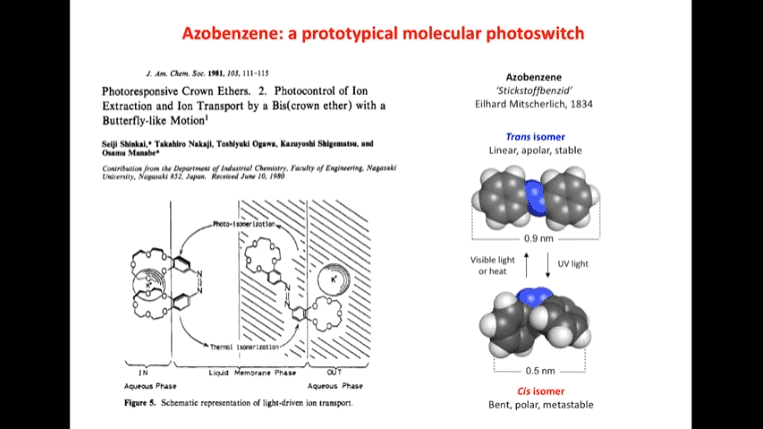
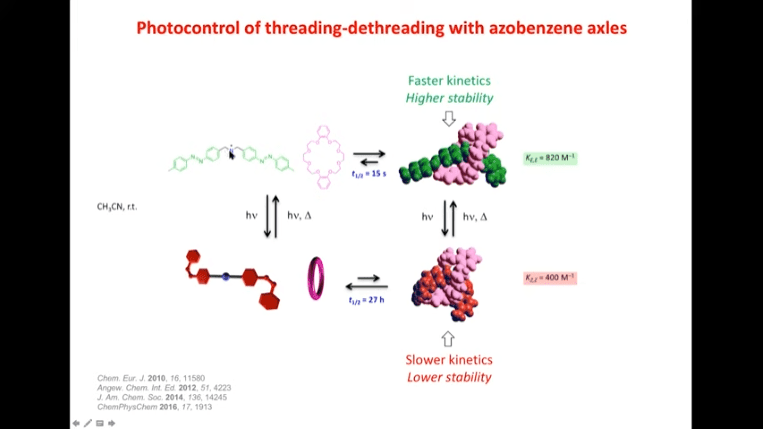
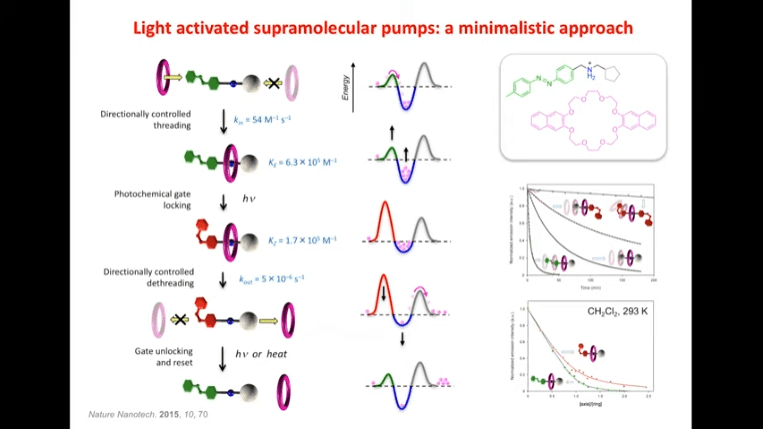
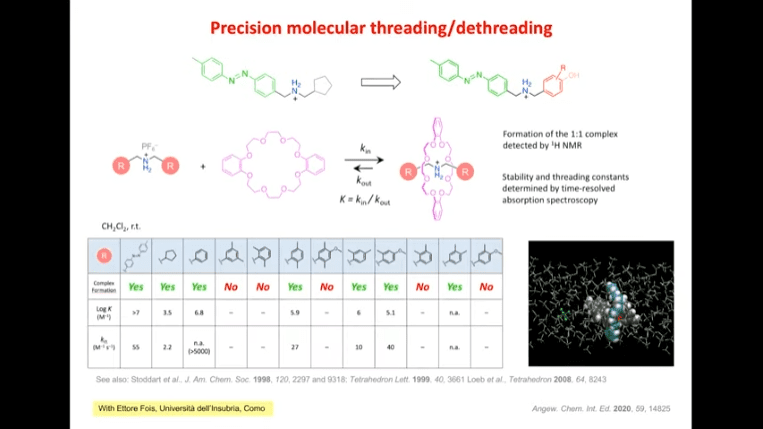
Rotaxanes are attractive molecules for molecular machines. The threading and dethreading process of rotaxane machines is crucial for their functionality. Light can be used to control the threading of rotaxanes by attaching azobenzene to the ends of molecular rods. Non-equilibrium states can be achieved by using light-induced rotaxane machines to drive reactions in one direction.
Opportunities
- There will likely be breakthroughs in the next 5 years in compartmentalization, organization on surfaces and interfaces, and systems chemistry. Further out, 15 years or more in the future – artificial life systems, a reinvention of catalysis, and a killer application that produces true value from nanotechnology.
Opportunities
- There will likely be breakthroughs in the next 5 years in compartmentalization, organization on surfaces and interfaces, and systems chemistry. Further out, 15 years or more in the future – artificial life systems, a reinvention of catalysis, and a killer application that produces true value from nanotechnology.
Opportunities
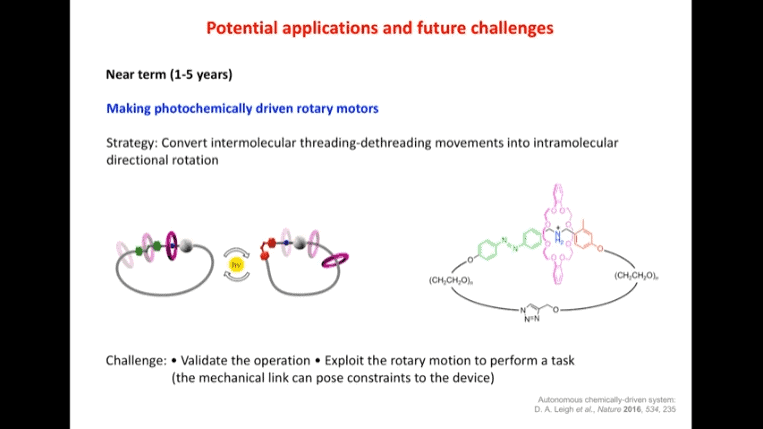
- In the near term (1-5 years), photochemically driven rotary motors are being investigated. There is also interest in intramolecular storage of light energy.
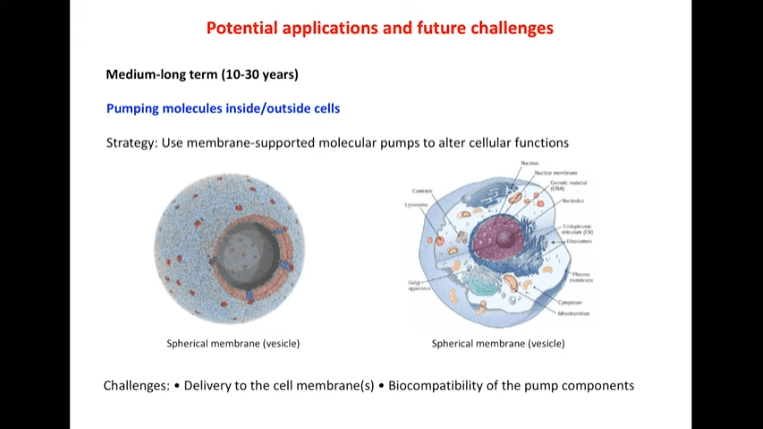
- The medium term (5-15 years) goal is the conversion and storage of light energy into chemical energy. Membrane and vesicle technology could be helpful for this goal.
- The long term (10-30 years) goal is to pump molecules in and out of cells.