Summary
In this session, Ronald Kohanski, the Director of Division of Aging Biology at the National Institute on Aging, presented his ideas and suggestions for a way forward from an NIA perspective. He covered many different areas in the field, starting with an overview of metrics and indices the field is using to track health, and the need for developing an aging scale focusing on health expectancy. Then he offered his opinion on interventions that seem promising, particularly on interesting research opportunities for heterochronic blood exchange.
Presenters

Ronald Kohanski, National Institute on Aging
Ronald Kohanski, PhD. is the Acting Director of the Division of Aging Biology at the National Institute on Aging, NIH. Trained as a biochemist, he obtained a PhD in Biochemistry from the University of Chicago in 1981. After a postdoctoral fellowship with M. Daniel Lane at the Johns Hopkins University School of Medicine, he held a faculty position at the Mount Sinai School of Medicine for 17 years before returning as a faculty member at Johns Hopkins….
Presentation: Promising Aging Approaches from an NIA Perspective
- In the face of diminishing capacity, are you still healthy? Yes, but the question is how do we understand that, how do we better maintain it, and for those whose health is declining a little bit more rapidly than we would like, how do we ameliorate that?
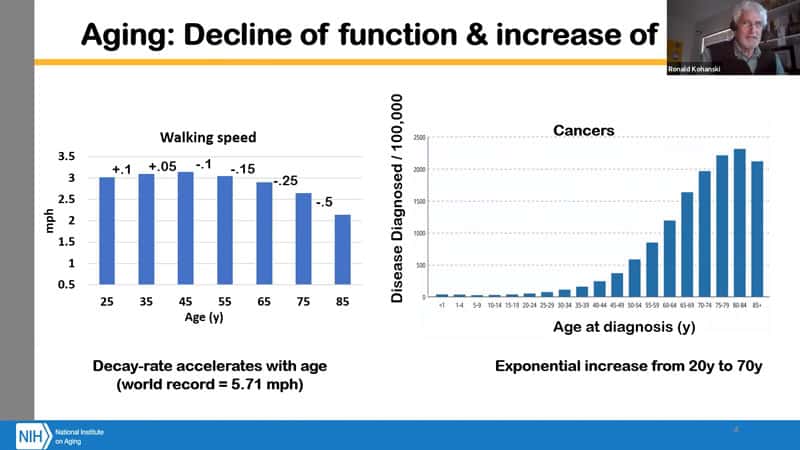
- Aging encompasses a decline of function and an increase of disease risk, which is basically what the geroscience hypothesis states. Aging is a major risk factor and if we could slow it, we could decrease the severity of the disease and delay its onset.
- Walking speed is a good predictor of mortality actually, and it decreases nonlinearly with age, reflecting the accelerating rate of aging (like cancer occurrence accelerates exponentially with age). A lot goes into walking, it’s a complex activity, and many of the components that contribute to being able to walk are measurable and possible to analyze.
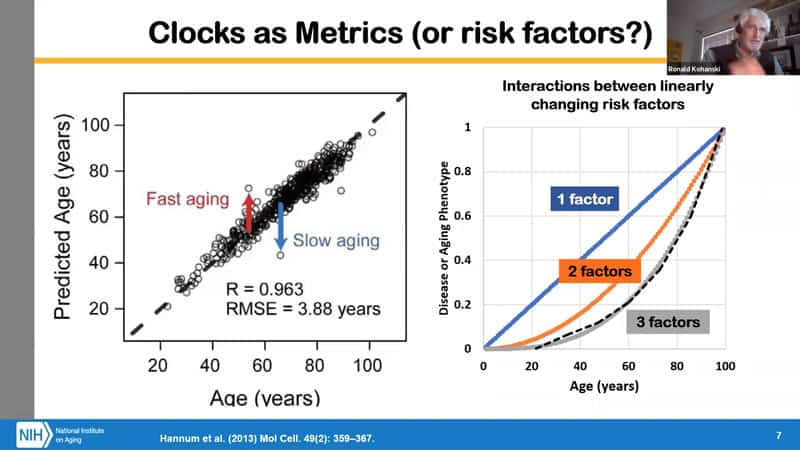
- Are linear clocks the rate of aging? Methylation clocks are looking for elastic net linear fit based on the methylome. It is interpreted as saying that those who have methylation age above the average set, they are aging faster, and vice versa, and it looks like the evidence is supporting that in a lot of caess.
- One of the problems is that methylation age is linear, but the resulting risk is exponential.
- Risk from a single factor may be linear. From two linear factors, you get a non-linear outcome if they interact. If you have 3 factors then it starts to look like an exponential change. It is as yet unclear how to relate the linear clock to the exponential increase in risk. It is likely that methylation clocks reflect at least one of the factors of aging.
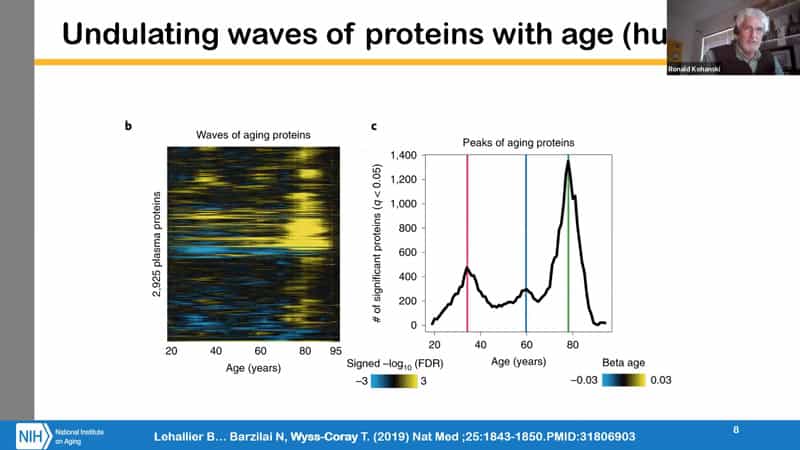
- Another important realization of the lack of linearity comes from the fact that we recently learned that there are basically three waves of change of aging proteins occurring early, late mid-life and in old age. What kind of clock represents this pattern of waves of aging is unclear as well.
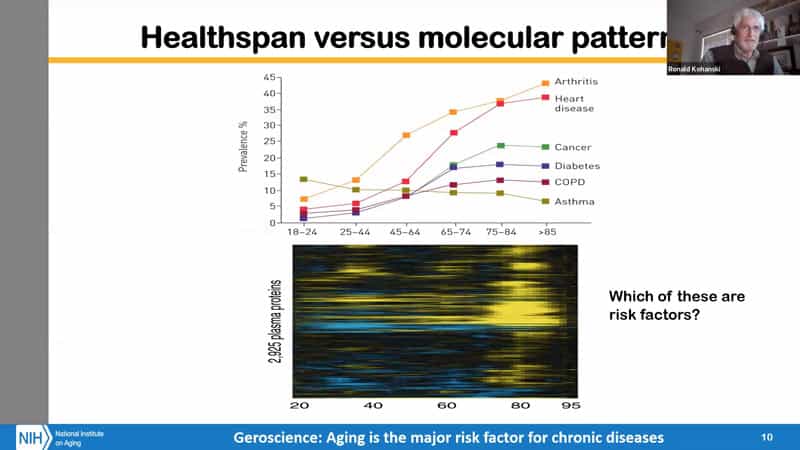
- The term healthspan became prominent because the metric we have been using for 25 years before was lifespan. We were pivoting towards health, therefore we got “healthspan”. But when we get into older ages, we get multimorbidities, which is why TAME trial that focuses on that is so important. Perhaps “personal health expectancy” will allow for good health to include well-managed diseases?
- The question is how do you relate all of these changes that you can detect in plasma proteome, methylome, metabolome, etc. to the prevalence of disease? How do we know which one of these 3000 proteins are risk factors, if any? That’s the challenge we need to unravel.
- There is another way of looking at it – a useful term might be health expectancy, which is already used in insurance companies, so they have an expectation how your health might look in the not so far future. Something like that might be very useful when it comes to geroscience. We’re asking for scoring health at older ages – biology or physiology – over time. Either you look at the recent trajectory or a single time point and then compare that relative to other known trajectories or collections of time points, and ask how old this person is relative to the rest of the population?
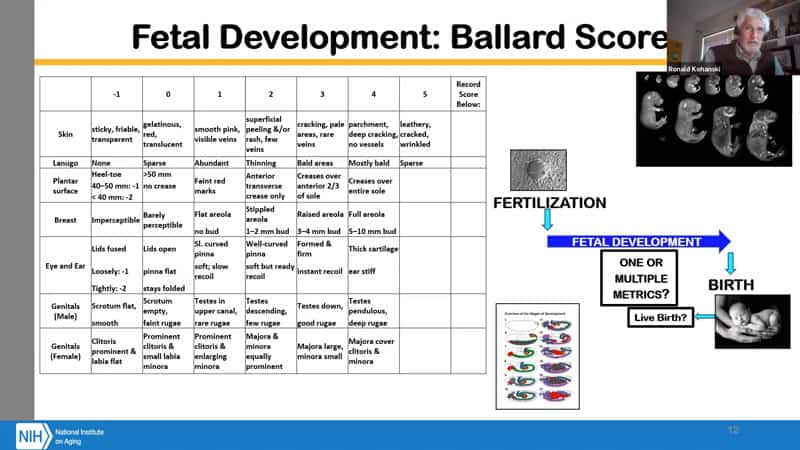
- There are lots of ways of looking at biology over time. There is fetal development for example – “Ballard Score”. You have a set of scores where you can look at a set of parameters and you can tell in which stage it is, but you can also identify the outliers and see when something goes wrong or fails.
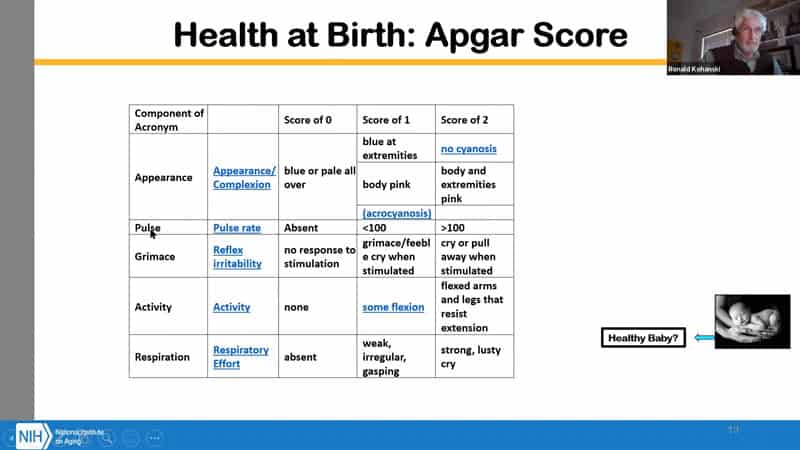
- Then there’s also “Health at Birth” – “APGAR Score”. Way of assessing a healthy baby with a score from 0 (inactive, without respiration, but can be treated) to 10 (things look pretty good), so there’s a score at birth.
- And then there are also sets of scales for what’s going on in puberty (Tanner Scale). And because it has multiple components, you can tell how well the person is doing in terms of maturation, and whether things are going in synchrony or become dyssynchronous. You can also see shifts in populations (for example, age at menarche), effects of environmental influences, etc.
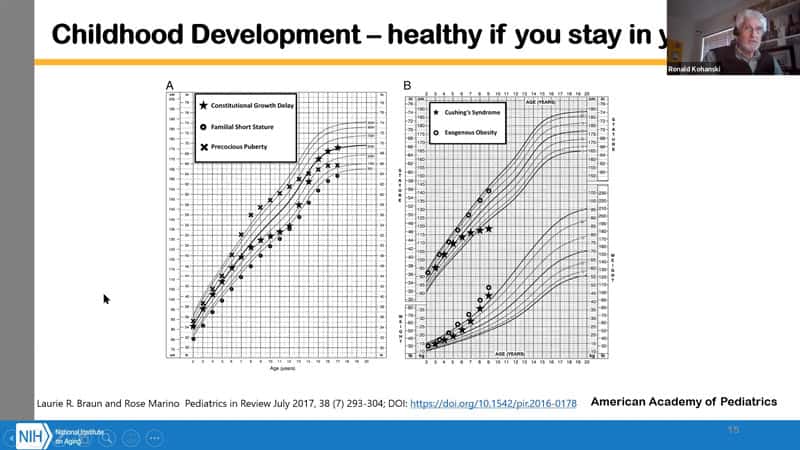
- We also know what constitutes health during child development. The American Academy of Pediatrics has these charts anybody can follow even at home. Basically you are healthy if you stay within the pattern- short and slim, tall and heavier, as long as your trajectory stays within limits, you’re healthy. But if you break across lanes, then that’s some indication of an unhealthy condition.
- So what do we have for aging? Interesting work is coming out from Vadim Gladyshev’s lab on that topic. But generally we could have an aging score with multiple axes and components – for example physiology components (resilience or frailty indices, some of them based on function, molecular characteristics, clinical characteristics, or combinations), extrinsic risks (which affect healthy age), network components (interacting networks and nodes affecting aging). The question is what kinds of outcomes are we going to predict? Is it “time to death” (which is useful, but could be disconcerting to be told), or is it “expectation for health”?
- The next step is to develop interventions – and we want to score them as well.
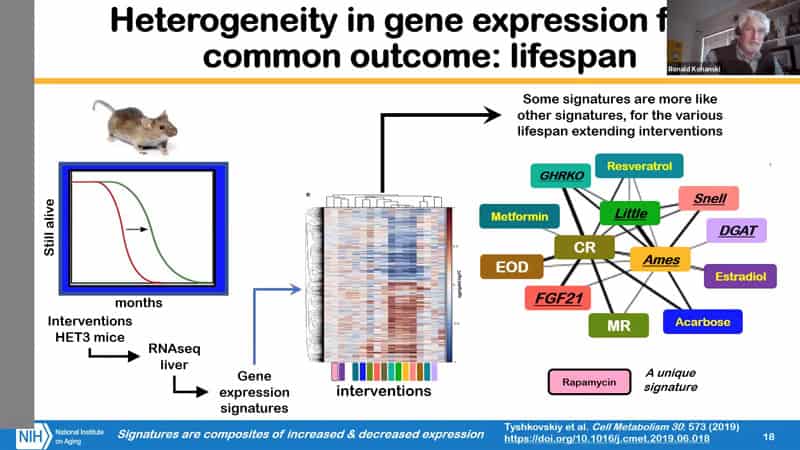
- One way to look at it is through testing laboratory mice. If you extend lifespan, the older mice tend to look healthier than the control mice. What the Gladyshev group did is that they took the liver from these mice and looked at the gene expression signatures in the liver. And then by clustering they’ve shown that some interventions have more in common than others. So then they’ve mapped these into a network space based on which we can find unique signatures that stand out, like rapamycin. The proposal here is intriguing, because if you can use this approach as a screen for an intervention that extends lifespan, might you actually have an intervention that extends health?
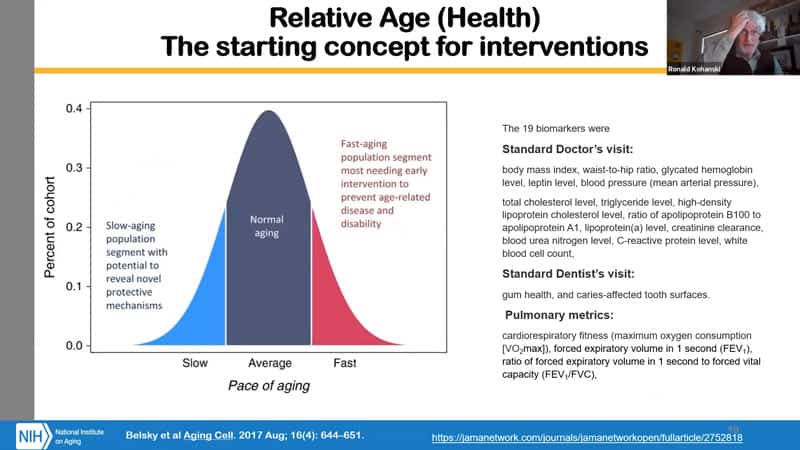
- If you look at relative age (often called biological or physiological), you get what you expect – for any population, you get people who seem to be aging slowly and who seem to be aging quickly compared to the average based on a set of characteristics. And it is apparent not just mathematically but even visually, if you take the people who are aging faster according to the biomarkers, they also look older when run through an age recognition algorithm.
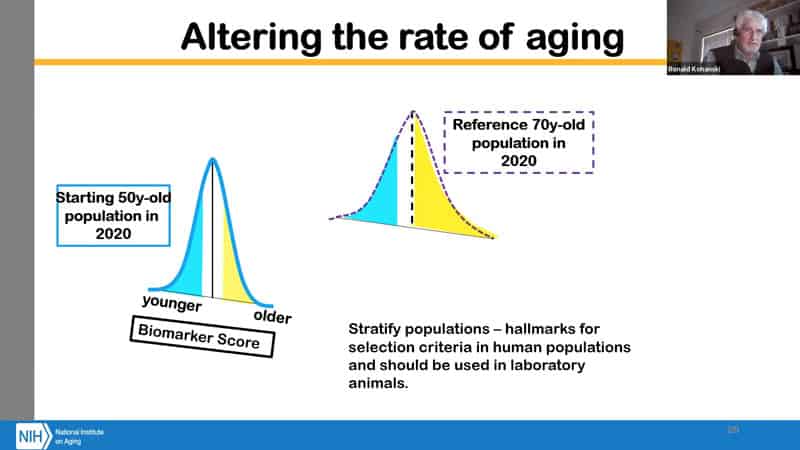
- Why is this a starting concept for intervention? If you have a population of a study that you want to do for let’s say 20 years, you can stratify the population based on the age recognition algorithm and therefore expect that the people who look older might benefit more from the intervention. So if we would use the whole population and look at the averages at the end, perhaps the averages would not shift that much relative to the reference population. What we want to change is the rate of aging, and see whether interventions can change the pace from old looking 50 year old to good looking 70 year old.
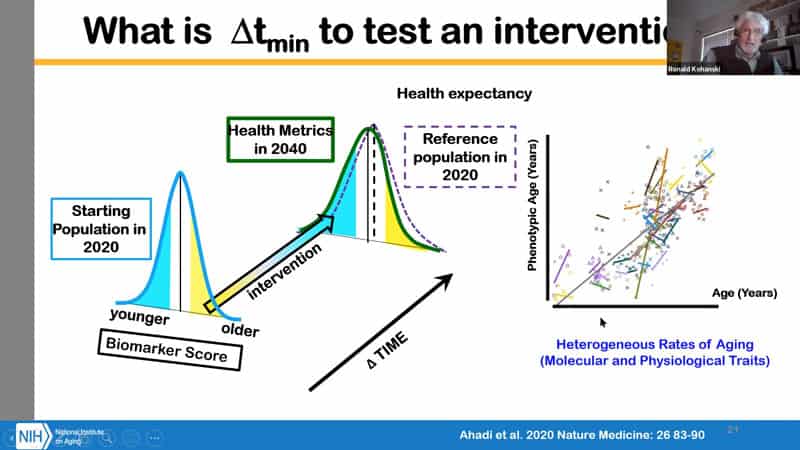
- Not everyone wants to wait 20 years to see if their intervention works. Aging is idiotypic, if we sample 20 metabolites for many people they won’t show the same patterns if they are aging at the same rate, that’s why we use the aggregate scores. What is intriguing about this study is that they’ve shown a way how to deduce or derive a rate of aging for individuals. Therefore in principle it should be possible to test interventions in much shorter time spans – like 3 years – through these ageotypes. And that’s encouraging, because it’s the same time span as studies with laboratory mice.
- A few organizing principles for interventions:
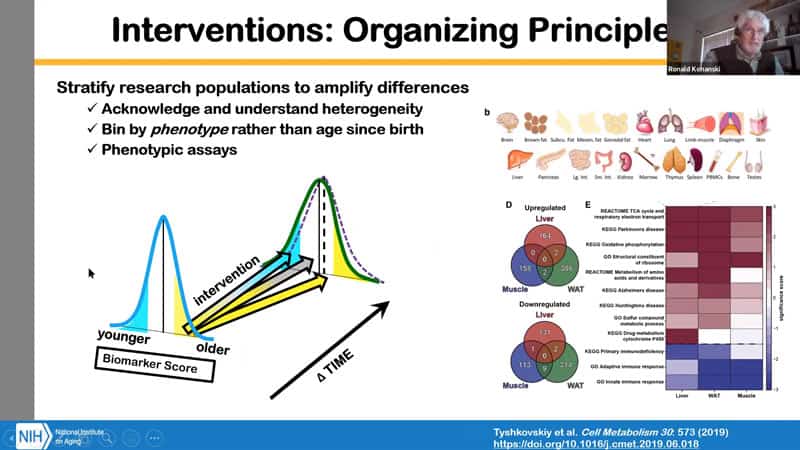
- We have to accept that aging is heterogeneous, at the organ, tissue, cellular and molecular levels.
- We can ‘bin’ within a population by phenotype rather than age since birth and that might allow us to amplify the signal and find out whether the intervention has strong impact, moderate impact, or no impact, or even unexpected or deleterious effect
- We need appropriate phenotypic assays (for various organs, and then compare
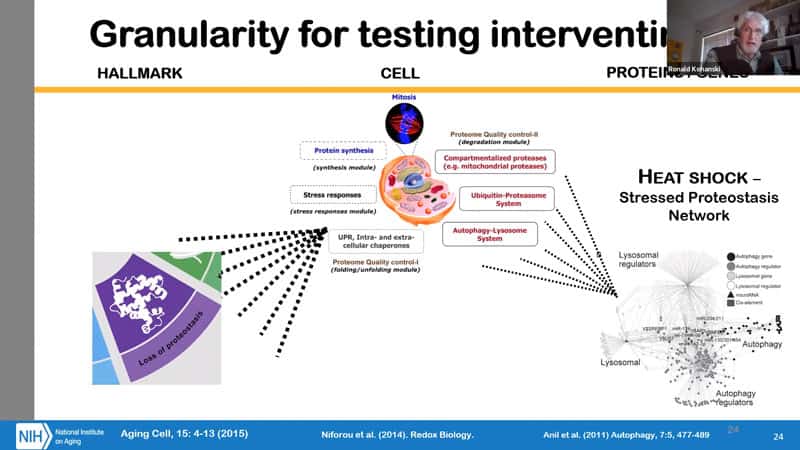
- There is evidence that dividing a population into subpopulations might be beneficial. In this case it is for loss of proteostasis, which is one of the hallmarks of aging. In principle segregating interventions can be quite informative.
- Another question when testing interventions is the granularity. Loss of proteostasis is one of the hallmarks of aging, and the proteostasis underlying that happens in organelles. And you can actually assay organelle function microscopically. Another way is to look at the very complex network underlying proteostasis. This whole spectrum is available in terms of what would be a useful phenotypic assay.
- Then we get to rejuvenation, as was discussed in previous amazing talk with Irina Conboy. They used an injury repair model.
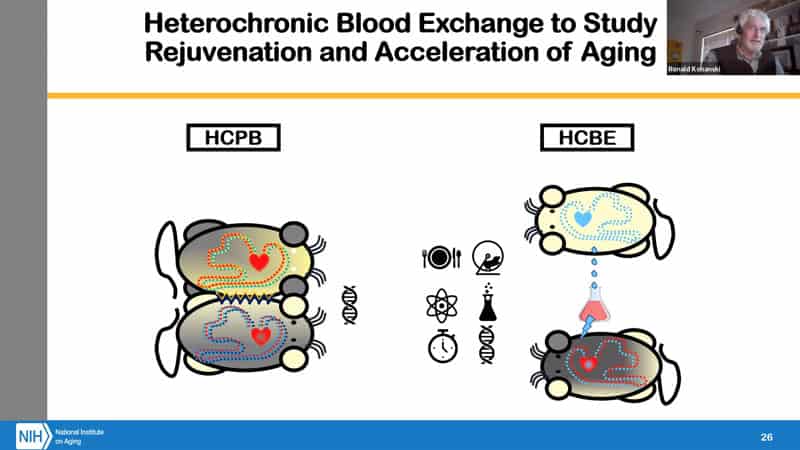
- But in other situations where it was not an injury repair model people have shown that when you conjoin a young mouse and an old mouse, that some of the features of the young mouse appear to rejuvenate the old mouse and vice versa from the old mouse to the young mouse. The immediate question was: “What is doing that”? We can learn about that through heterochronic blood exchange that Irina developed. You can do all sorts of things to one of the mice, then take the blood from it and put it into other mice without the intervention through heterochronic blood exchange. And see all sorts of effects from different interventions perhaps better than through parabiosis.
- One of the things we can ask about rejuvenation is what is necessary and what is sufficient?
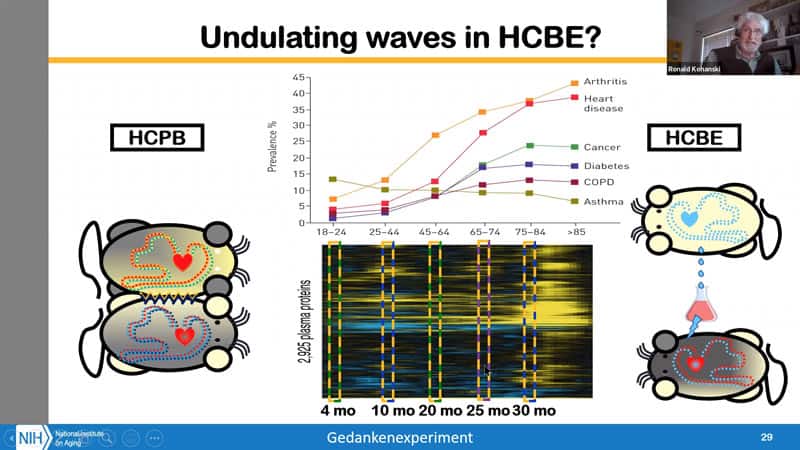
- Let’s do a thought experiment and try to superimpose the undulating waves of plasma proteins in HCBE. We could map the age when the waves of plasma protein changes happen in humans to the mice age. And then we should figure out whether the researchers just got lucky in picking these two ages and finding the transfer of aging phenotypes, or whether we could through HCBE investigate mice and their different expression profiles before and after the different waves of plasma protein changes, before and after the advent of disease state, to see whether there isn’t any more information in there. Have we just got lucky with this experimental paradigm and is there more to it that we have not yet leveraged? And of course
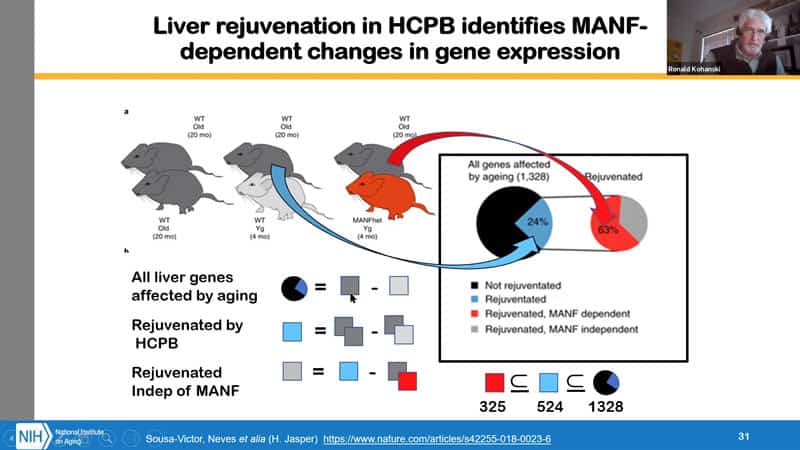
- You can see different effects when you join two old mice, young and old, and old with model mouse for this specific experiment with lower MANF.
- So what’s laid out here is a way to analyze this experiment for causal connections, because the genotype has been altered at least in one gene. And if you look at all other genes affected by aging, that’s young vs old without the conjoining. Then you have those that have been rejuvenated by HCBE, and that’s what you get when comparing data from the old vs old and old vs young. And then the genes that still change in the pair with model mouse with lower MANF, those can be considered relatively independent of MANF because you’ve reduced the signal from MANF. So then you have a smaller set of genes to work with that provide the clues to mechanisms for liver rejuvenation.
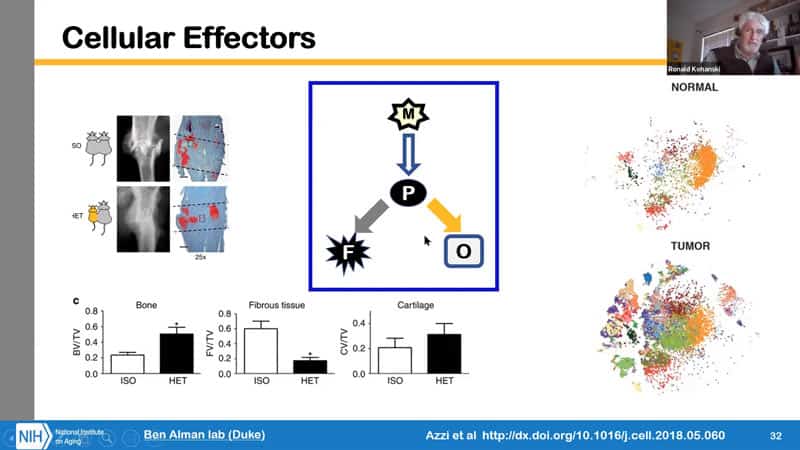
- Now we can look at the repair injury model instead of proteins. We know that fractures in older people and animals do not heal as well. What was shown in this study by Alman is that macrophages move into the fracture area and affect progenitor differentiation, so they affect a cell fate decision. Interestingly enough, using gene expression profiles, they have been able to identify which are those macrophages, what is their level relative to age and what are their actual characteristics and their organ of origin and also their developmental origin.
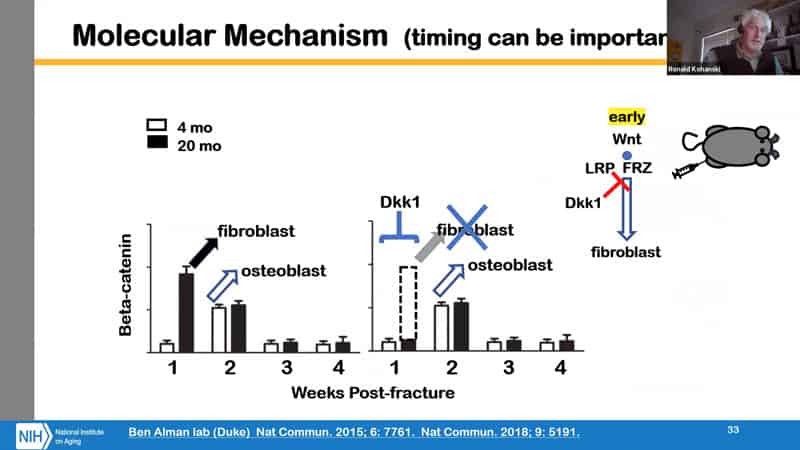
- What they also did is this other thing, when you’re dealing with rejuvenation in an acute situation, we might expect that timing can be important. We know that when doing chemical or pharmacological intervention after a heart attack, the sooner you get to the patient and give them the appropriate pharmaceuticals, the better of the patient will be. On the other hand for cell therapy, an interesting observation was made that there is a better result if you actually wait a little bit longer, since there were also cells coming through bone marrow signaling from the injury, which takes a few days, so delaying is beneficial in that case. So we could do a two regimen intervention, one short term with the pharmaceuticals and then long term repair using cell therapy. On the slide is an example of timing importance at the molecular level with the WNT pathway.
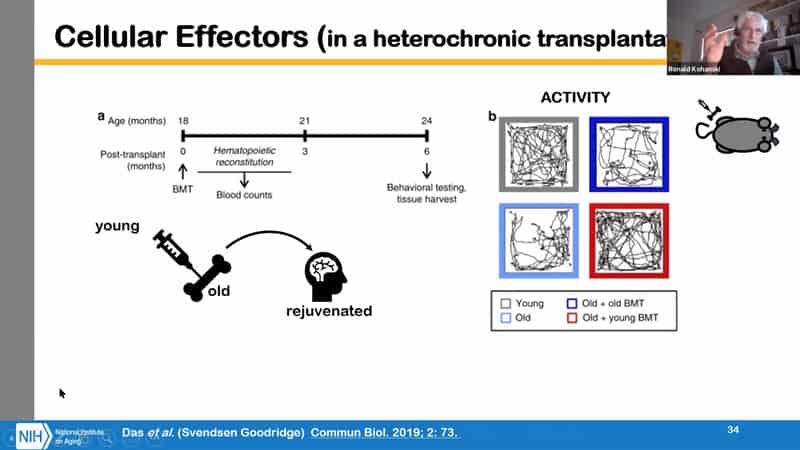
- This is another example of cellular effector moving through the blood. This was bone marrow cells taken from young animal and injected into the bone of an old animal. And what was rejuvenated was cognitive function along with activity. So this is a phenotypic screen that could be useful.
- The NIA just did another request for applications for HCBE, 9 new projects are going to be funded. Details about the specific project will be released later, but in general what is intriguing about this set of applications is that they are uncovering the origins of factors that will be rejuvenative, coming from all over the body, from all sorts of tissues, affecting many various targets. It’s remarkable what this kind of experimental paradigm is able to do.


Nathan Shock Centers program was renewed, it’s been going on for like 30 years already. Multiple labs covering multiple topics on biology aging with all sorts of great imaging and all sorts of great technology, they work with laboratory animals, human populations, human materials. They have a lot of technology and collaborations could be arranged with centers or investigators themselves. One of the leading centres is at Albert Einstein’s College of Medicine under Nir Barzilai.
Q&A
You mentioned it’s easier to do things when you’re healthier, and we know that exercise itself has a geroprotective effect. So it stands to reason that the eventual beneficial effects of an intervention could be split into first order and second order causes (like the biological effect of the intervention, and how it made you feel and perhaps exercise more, etc.). When it comes to having to rank interventions to rank first order effects, is there any system in place to normalize or tease out these two potentially compounding components? By normalizing based on mobility changes or anything like that?
- That’s a sort of question that a trialist would be more able to answer but I can speculate a little. You track what’s going on and look for secondary effects and if they feed back, they can be treated as confounders of the primary outcome. And there are actually graphical and mathematical ways to display that and tell you whether something is significant in terms of relating the intervention to the outcome or if it’s a modifier of the outcome..
Nir, is that kind of analysis planned for the TAME trial, to normalize for the mobility increases and things like that?
- Absolutely, 25% of the budget is a grant from the NIA to take all the biomarkers from TAME and make sense of them. Because what we’re missing mainly is not biomarkers, but the knowledge about which biomarkers are changing with aging and which to therefore track, so we can look for changes in shorter periods than years-long trials measuring outcomes. You have to plan it very carefully and do all the analysis to really understand what’s going on.
- You’re raising a very good point though, because what you’d like to get from intervention is for people to be able to do more of the things that have beneficial effects. You want to have some ways to increase their independence.
Who are the other champions within the NIA who are supporting this direction of research? Or are there any opponents?
- We’re all in this together, we collaborate quite a lot. There are multiple divisions (geriatrics and clinical gerontology, behavioral and social research, neuroscience research). We also work together with other NIH institutes. Pretty much every other institute is now paying attention to aging as a parameter. And there’s also a NIH wide approach to include older people in clinical trials and research. There is not really opposition, more enthusiasm, with due caution because of the problems associated with asking older people to participate in clinical trials – there are serious ethical concerns in some cases when they are cognitively impaired.
Is anybody at the NIA doing work specifically with imaging data in relation to predicting aging biomarkers?
- Yes, there’s a lot of work being done with imaging technologies. We actually had a project for imaging aging, and we do have similar programs in the area of validating biomarkers for clinical trials. For example, cell painting that reveals about a 1000 of cellular phenotypes, cryo electron microscopy, work on chromatin structure, and imaging from dexa scans.
If you can pick from the entire universe of biomarkers, what was the most predictive of the rate of aging and future longevity?
- I don’t have one favorite biomarker. There are many projects going in the right direction, including the work done by Steve Horvath and Morgan Levine.
How can people learn about new grants from the NIA?
- Funding opportunities are decided by council advisors in January, May, and September and visible on the NIA website in the recordings of those meetings. The next meeting is in mid May. Watch the website.
- You can also file a request for information (RFI) with NIA’s abbreviation to look at funded work in the past.
- You can contact anybody at NIH through [email protected].
Are there going to be any changes in the structure of NIA or NIH that are relevant that you can talk about?
- Expanding focus on health disparities in relation to aging and increased focus on research in human populations. We have around 70 grants with human populations. The technology to do basic research with humans is available, so we should be moving that way and are trying to do so.
What challenge would you like to see more work to be done on?
- Widening our understanding of basic biology of aging in the human population. This includes widening and diversifying the human demographics we study and finding promising intermediary candidates between mice and humans to study aging.
What does NIA want more of, what can people help with?
- Public private partnerships: partnering with industry on projects of common interests.
Seminar summary by Bolek Kerous.