Summary
Irina Conboy from UC Berkeley presented the latest findings about tissue rejuvenation via plasma dilution that has major effects on aging related processes. Following that is a discussion touching on theoretical and practical next steps, limitations, including information on the most likely path to market and possibility of availability for the general public in the relatively near future.
Presenters

Irina Conboy, UC Berkeley
A key direction of my laboratory is to understand age-imposed and pathological changes in molecular compositions of systemic and local environments of adult stem cells and to calibrate these to health – youth. In the past few years this direction has been ramified into…
Presentation: Irina Conboy
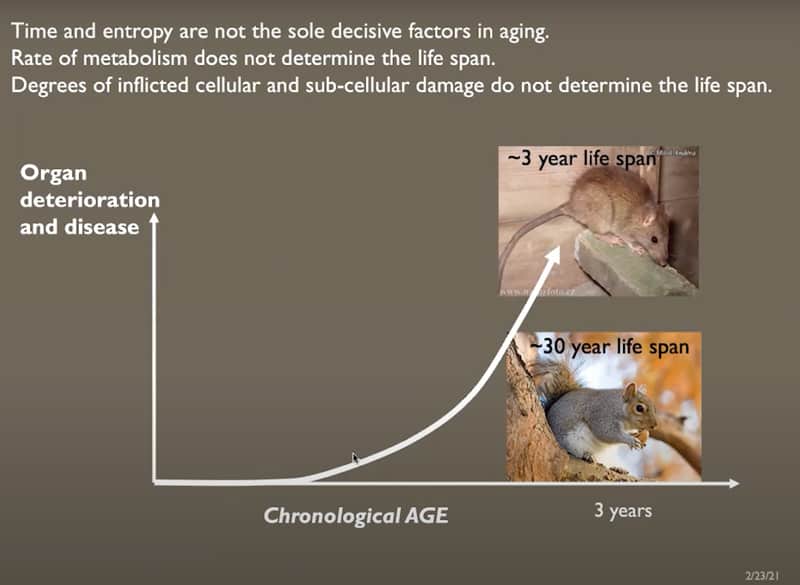
- Time and entropy are not the sole decisive factors in aging. Rate of metabolism and degrees of inflicted cellular and subcellular damage do not have to determine the life span (rats live ~3 years, squirrels ~30 years). Life / health span is controlled by the efficiency of repair / regeneration.
- Axiom: When several key old organs become significantly younger and healthier, the old mammal is younger and healthier.
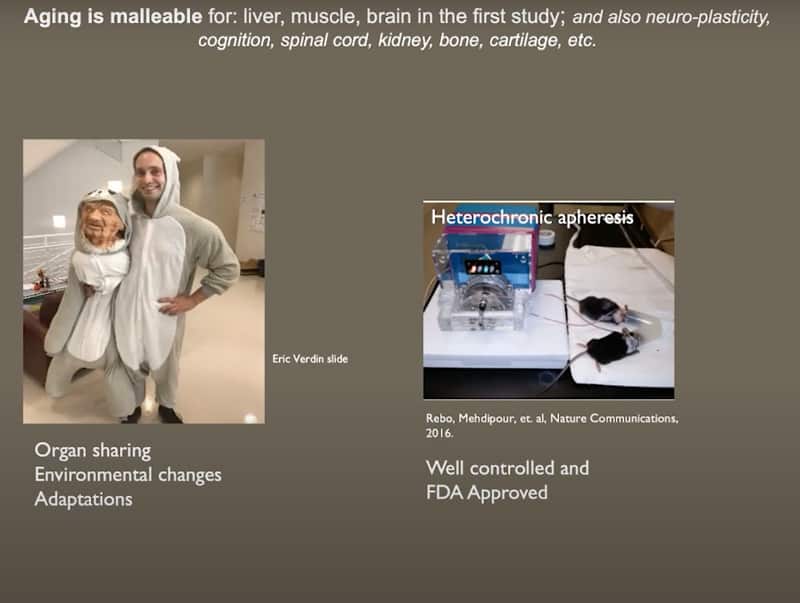
- Based on parabiosis mice studies, a more controlled process for mice experiments with blood exchange was developed; heterochronic apheresis. It was demonstrated that aging is malleable for: liver, muscle, brain and neuro-plasticity, cognition, spinal cord, kidney, bone, cartilage, etc. Compared to parabiosis, this is also better translatable to the clinic, because plasmapheresis for humans is already FDA approved.
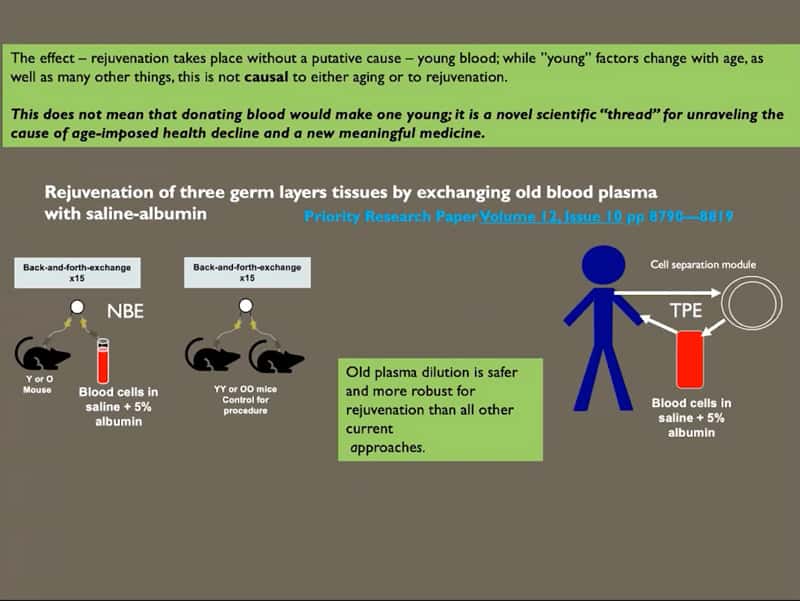
- The discovery was not about young blood but a proof that mammalian aging is systemic and can be prevented, attenuated, and reversed to youth. Rejuvenation takes place without a putative cause of young blood. The next step for the “young blood way of thinking” is focusing on exercise-induced rejuvenation factors or a combination of dilution of old blood plus adding positive aspects from young blood.
- Therapeutic plasma exchange / plasmapheresis / old plasma dilution has the potential to be safer and more robust for rejuvenation than all other current approaches.
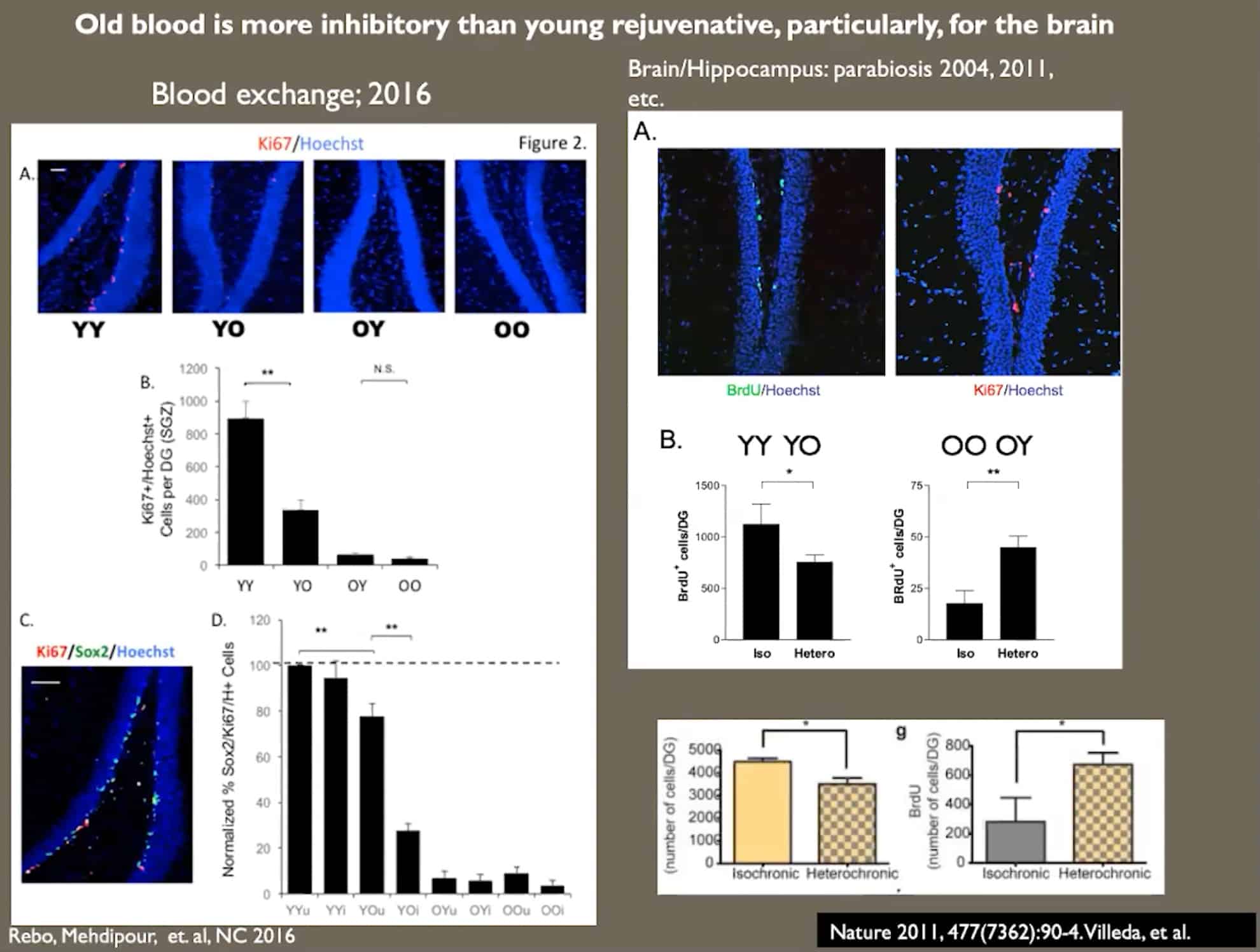
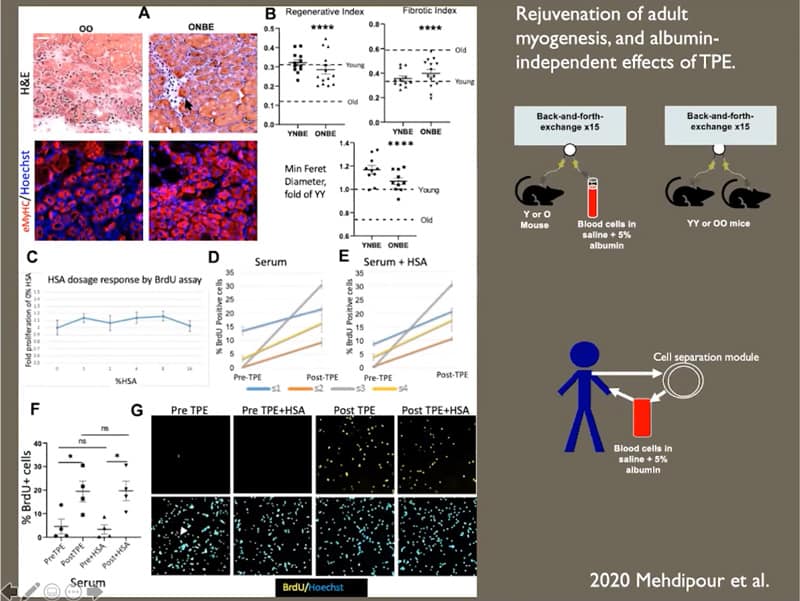
- Blood serum of old mice is inhibitory for stem cells, which explains why their regeneration declines and why plasma exchange improves it.
- It doesn’t seem that albumin is the main factor in the system, there was no dramatic change in effect when albumin was not included.
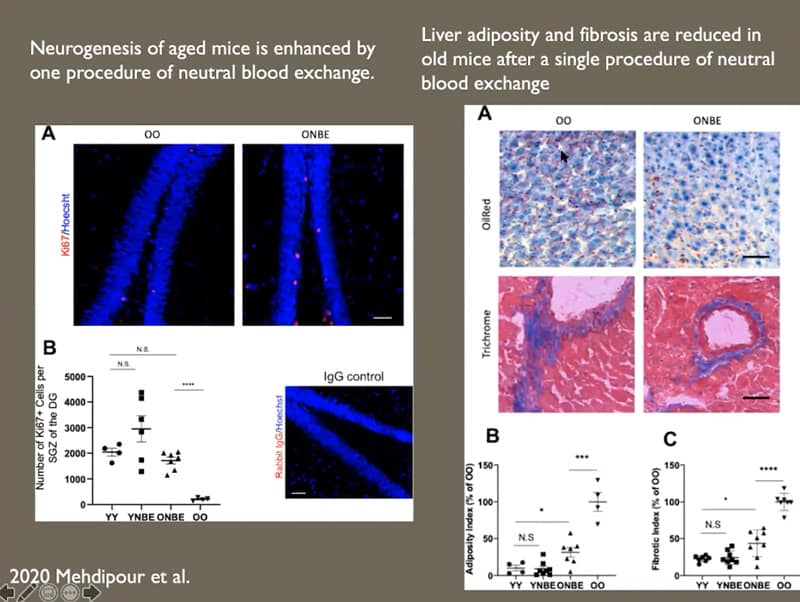
- Neurogenesis in old mice is enhanced by a single procedure of neutral blood exchange.
- Liber adiposity and fibrosis are reduced in old mice after a single procedure of neutral blood exchange.
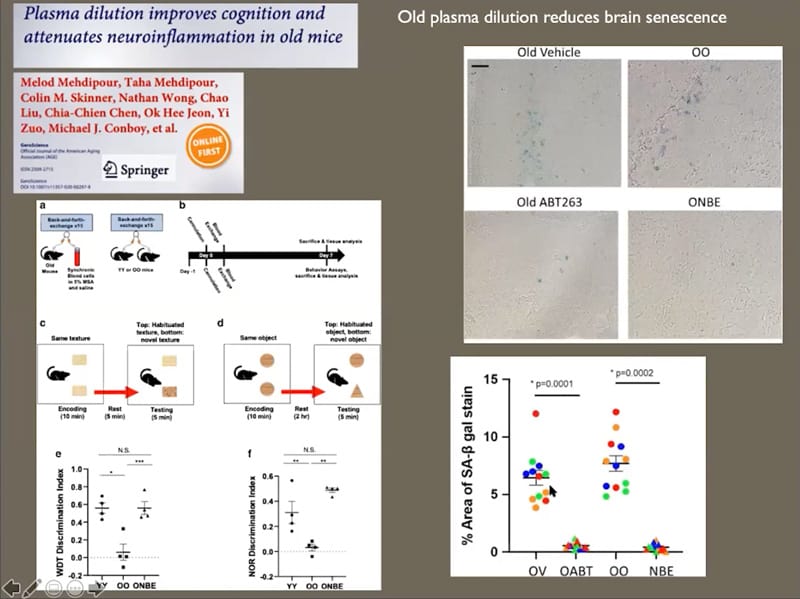
- Old plasma dilution reduces brain senescence, neuroinflammation, and improves cognition.
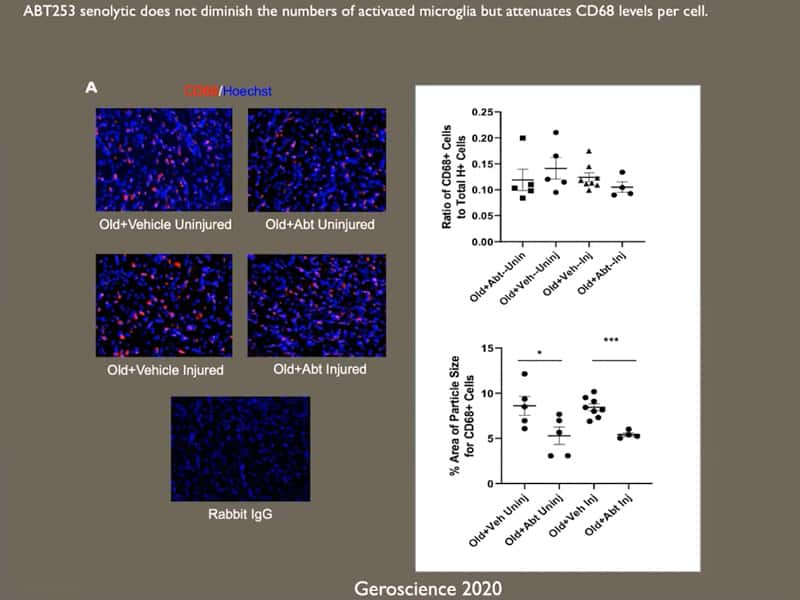
- Neutral blood exchange seems to have senolytic or senomorphic properties, so in theory it might not be necessary to use senolytics when you use neutral blood exchange.
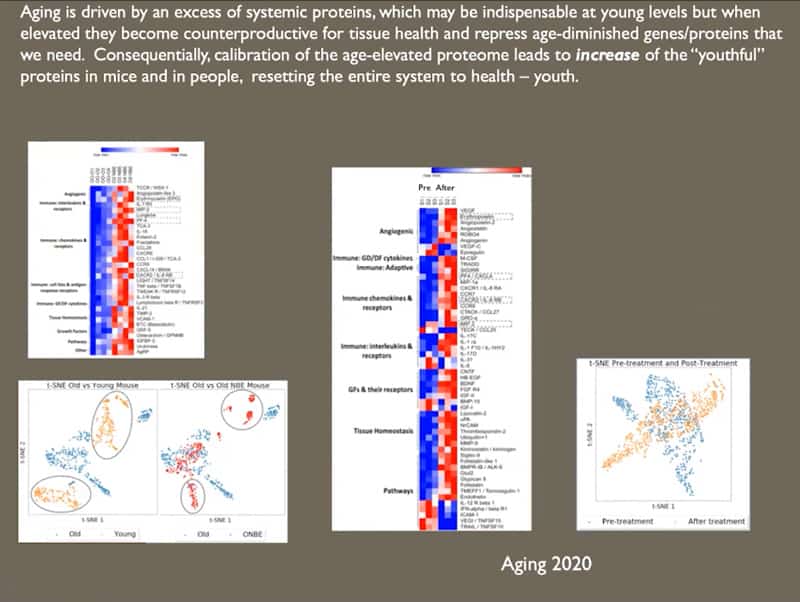
- Aging is driven by an excess of systemic proteins, which may be indispensable at young age and levels, but when elevated they become counterproductive for tissue health and repress age-diminished genes and proteins that we need. The youth proteome is basically repressed by the old proteome. Consequently, calibration of the age-elevated proteome via dilution leads to an increase of the youthful proteins in mice, resetting the entire system to health and youth. This happens in two waves of rejuvenation, the first wave comes from the dilution of repressive factors and normalizing of age-related proteins, the second wave comes automatically after that when the youth proteome is restored because it is not repressed anymore. And that results in enhanced tissue maintenance and repair.
- The conclusion is that large dilution of systemic milieu stably resets molecular, cellular and tissue determinants of organs’ health and repairs it to younger states. This makes all or most old organ systems rapidly and robustly younger.
Q&A
Scientific questions
What are the limits of this plasmapheresis procedure in terms of rejuvenation, if there are any?
- There is currently a clinical study in progress aiming to get a better understanding of 1) how long does one procedure last (what is the duration of positive effect and how often does the procedure need to be repeated) and 2) how many age-related diseases can be tackled (TPE is not approved for age-related diseases, so we need to have a study that shows which age-related diseases are affected, and how many hallmarks of aging can be offset). Based on that data we will have a better idea of the limitations.
What are the assumptions about the mechanisms of neutral blood exchange affecting senescent cells – is it that there is improved immune clearance or that the cells are becoming “de-senescenced” back to normal?
- This is yet to be determined and there is a challenge with the measurement markers of senescence. But the hypothesis is that the cells are becoming “de-senescenced” (unlike to how senolytics work).
Based on the proteomic data, is it possible that you are actually rejuvenating the immune system? And could that be one of the reasons why you’re seeing the loss of senescent cells? And as a final corollary of that, do you know if your animals have better resistance to infectious diseases?
- Immune regeneration is the main aspect of this TPE approach. And perhaps it is also another reason for better clearance of senescent cells. Not just animals but even people that are now undergoing the clinical trial have improved resistance to infectious disease.
How much faster do young mice get worse when given old blood?
- The liver and brain very quickly, in one week after one exchange. A single 50% exchange has a huge effect, even with young organs. But that doesn’t hold for muscle for example. Some organs are much more fragile and prone to the effects than others. Also interesting link with injuries, if the muscle is injured, then the brain becomes old much more robustly – muscle injuries make the brain more fragile. Could be because of increased inflammation in the liver and kidney, from which then the brain suffers.
What are the assumptions behind aging being driven by an excess of systemic proteins vs. the excess of systemic proteins being a downstream consequence of other drivers?
- There is for sure some chronology and general cascade before excess systemic proteins happen. The excess of systemic proteins is initially caused by damage to cells and tissues. But because the proteins are systemic, they then propagate aging and senescence to other tissues. The whole cascade itself is most likely not preventable because the initial damage is caused by things like oxidation and mitochondrial activity, and we cannot stop eating and breathing. This is also why intervening in the cascade at this step and addressing the systemic proteins makes sense.
Have you considered looking at additional features of brain aging, such as the “opaqueness” of the brain as the measure of protein damage or protein aggregates that is possible to observe with two photon microscopy?
- There is ongoing collaboration with a laboratory at Stanford, which should be looking for more brain-related data (using two photon microscopy for example) in further studies.
- It is not likely that a single session will undo a lot of this accumulated damage right away in the brain, but that’s where the two waves hypothesis comes – that the first wave will remove the old proteome that is continually forming the aggregates and enables the young proteome to come in and gradually improve repair and regeneration.
It seems like there are a relatively small number of CRITICAL negative factors (a dozen?) despite the fact that hundreds of factors may be involved. Do you see additional advantages of depleting negative factors specifically (with affinity column) versus dilution of all factors?
- Typically there is a hierarchy of dominance of interactions of key factors and determinants, which basically makes these networks that are controlled by a few key present factors that determine all the others, so going after a few key factors is the strategy, because renormalizing them will take care of hundreds of other factors.
Do you think there is any molecular weight cutoff of the molecules that you think need to be removed?
- No, there are age-related proteins with very different molecular weights, so it’s not possible to simply filter based on that. That is also why it is not possible to find a single fraction that will make you younger.
What is Irina’s key challenge for the Foresight group and accelerator?
- Safely calibrating circulatory milieu to health and youth. The way forward is through removal and dilution of age elevated proteins, and we need to have excellent screening systems and excellent computational and modeling capabilities to identify every single determinant age-elevated protein and to see how to calibrate them to precisely young levels. However not just in general, but individually. Because people do not age identically, so even though there will probably be the same patterns in our aging, there will also be deviations in those patterns between people. So the safe calibration should be precision based on patient level, where for every person we will identify the key proteome, the levels, and composition of it, how it differs from the healthy and young proteome, and how we can reset it to that healthy and young proteome.
Commercial questions
Can you give us an outlook on what the next steps are (with the research in the university and with the company)?
- In the university, they are wrapping up the scientific results of the 25-40 people clinical trial, which should shine a better light on the weaknesses and strengths and limitations of the procedure for treating human aging and age-related diseases.
- As for the company, they want to launch it probably in May when the results of the phase 2 trial are published and at that time get funding for placebo randomized phase 3 (right now the studies are longitudinal without a placebo group control), as well as funding for the branch of the company that will provide this service for a fee.
What is the high level thinking on why you can commercialize it since it is already approved, what can you protect with the company in terms of IP?
- TPE is already approved for only some diseases. The IP protection will be for a trade secret, because there is a combination of tweaks to the regular TPE process with the procedure. Another possibility is to re-patent for a new application of a previously approved patent. And of course there are numerous avenues for changing the composition of exchange fluids such as the novel chemical composition itself is IP protected. Those are the possibilities for the near future and for the long-term there is the possibility of patenting the process that safely identifies for each person the precise combination and concentration of things to be removed and to be restored.
The procedure sounds fairly low risk. Can people get this procedure now already somewhere?
- There are some risks, but they are much lesser if you compare it to injecting somebody else’s young blood. Yes, some MDs already offer TPE. For example the clinical collaborator in these clinical studies, Dr. Dobri Kiprov, has 25 years of experience with it and made a few innovations to make the procedure better.
What do you think about existing clinics in Europe that do plasmapheresis?
- Some people are already getting in touch, so there is an affiliate program under development that will make it so these affiliated clinics under the training of Dr. Dobri Kiprov propagate the know-how more broadly in the EU and USA.
How can people in this group do that would particularly help with the company?
- Mainly networking and advice in the commercial realm, as well as paths for resources for phase 3 clinical trial. That is the main priority so FDA can approve the procedure for new classes of age-related diseases and advertise it broadly as such, and perhaps have health insurers involved in compensating some of the resources. So resources and networking are very welcome.