Summary
In this session, Christopher Leptak, the Director New Drug’s Regulatory Science Program Office in the FDA’s Center for Drug Evaluation and Research, elucidated how is FDA approaching biomarkers specifically for drug development, what is actually considered a biomarker and wha needs to happen for a new biomarker to be used. Apart from valuable tips and insights about the process, and range of resources on how to prepare for it, he also offered his opinion on the way the longevity field could approach biomarkers with relation to aging.
Presenters

Christopher Leptak, CDER Biomarker Qualification Program, FDA
Christopher Leptak, Director of the CDER Biomarker Qualification Program at the FDA, will join us for a discussion on how to develop biomarkers for FDA-recognized diseases. While aging is not an FDA-recognized disease, and thus falls outside of the scope of this discussion, Christopher may help us shed light on the…
Presentation – Christopher Leptak
- With all the digital biomarkers popping up, the definition is becoming a little bit fuzzy in the public discourse, but the FDA has clear criteria for biomarkers.
- Important resource called BEST – long-term effort between NIH and FDA to collect a glossary and codify terminology within the biomarker space. It’s a living document that’s updated periodically trying to ensure that everybody can be on the same page.
- We try to define biomarker classes around their use as a way to differentiate them between each other – the 7 classes are listed above.
- But it’s probably easier to think about different types of biomarkers through this flow diagram.
- “Normal” physiology – For a given individual, a normal doesn’t mean unchanged, it means within defined parameters. For a given person, throughout the course of a day, or a period of time, depending on the characteristic you are looking at, it might change. And that normal variability is important, because if you wanna say that change is important and wanna make a regulatory action on it, you need to know what the level of change is with respect to normal variability. So within the normal states some of the biomarkers we are looking at are susceptibility or risk where you try to predict a likelihood of disease in the future even though at present you might not have clinically manifested signs or symptoms.
- Pathologic changes – The changes from normal variability can over time result in pathologic changes.
- Altered physiology – Those pathologic changes can lead to altered physiology.
- Clinical disease – And the altered physiology is leading to clinical disease. The FDA approves drugs based on a disease, and that group of diseases also changes over time. Within a clinical disease, once you have that definition in play, there are few classes of biomarkers, namely diagnostic, monitoring and prognostic.
- Diagnostic can fall into two camps – 1) the biomarker itself can be necessary and sufficient for the diagnosis of a disease where you don’t need any other information (we call that the capital D), 2) but the majority of biomarkers are (lowercase d) that along with other information and biomarkers and clinical signs and symptoms as a “gestalt” define a clinical disease, or even a disease by exclusion. That’s certainly a relevant class of biomarkers to help your patient population for a clinical trial.
- Monitoring is particularly helpful for a condition that has some time element to it (chronic or not just acute but somehow active in a longer time span). In such cases you might want to monitor and look for the disease activity, since some diseases are going to wax and wane in nature, and you want to enroll populations that have active disease in the clinical trial so you see whether the therapy is going to mitigate that or not.
- Prognostic class is important because in a clinical trial we ultimately want to look for a clinical benefit. And so you want to go into your trial enrollment and enrich for patients that are likely to have that clinical event of interest within the time of the clinical trial. If the clinical event of interest might take 10 years, then the biomarkers are going to be helpful to enrich the patient population and maybe even look for an earlier event than that 10 year outcome.
- Change of physiology – When a therapy is in place we look at the remaining classes of biomarkers, ultimately here we want to show some evidence of engagement. Pharmacodynamic basically says that the therapy has some biological effect. But it doesn’t say that the effect is either beneficial or detrimental. So that can be proof of concept or looking at the mechanism of action of your product.
Predictive help to take a patient population and subcategorize them to say “this group with this particular feature may be more likely to have a positive effect or more likely to have a side effect and be a safety concern”.
Safety is another way we monitor the effect of a therapy on a patient, because we know that every drug, even the OTC drugs have some safety concerns.
- Non-progression or reversal – Ultimately what we’re trying to do is to change the physiology and the clinical course of disease so we no longer have progression or perhaps even have reversal. This is where we get into the response part of the spectrum, which themselves can become the endpoints, and which are measuring non-progression or reversal of the disease.
- Improved clinical benefit – And a subset of those that are predictive of clinical benefit are surrogates. Surrogates can become the basis for approval be it accelerated or traditional.
- At the bottom is an example of one of the qualifications of COU a few years ago.
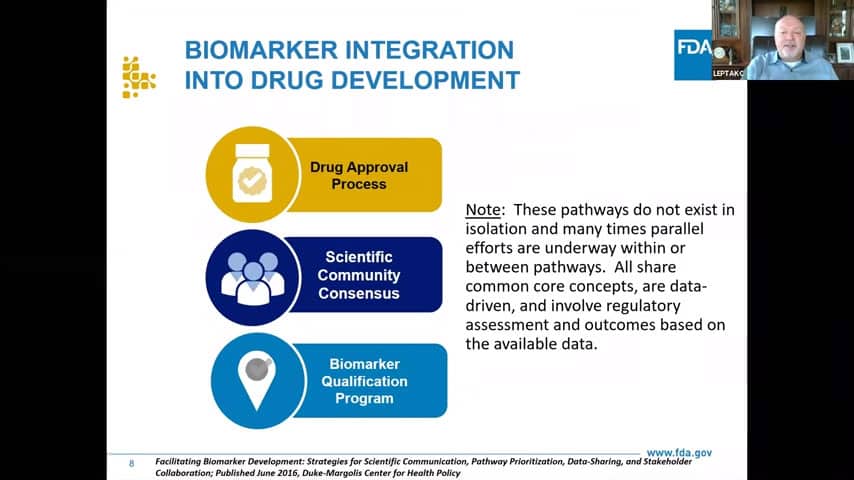
- Biomarkers come into the agency from many different places:
- Individual companies are bringing them under IND within the drug approval process, from where historically most of the biomarkers came and remains to be the biggest area.
- Then there is what we call the scientific community consensus – all the publications and scientific journals, professional societies and their position statements. This category is a great place for hypothesis testing, from where people can bring these hypotheses and bring them into either a drug approval process or a biomarker qualification program with all the evidence.
- Biomarker qualification program, which has been around now for over 15 years already, and has been formalised as a part of the 21st Century Cures Act in 2016.
- Do know that biomarkers are used and information lookup can come from each of these, we don’t have a preference. They are all data driven, on our side of the fence we have multidisciplinary teams that look at the information and many times biomarkers are developed in multiple categories simultaneously, which is helpful for us because we can find information from multiple sources.
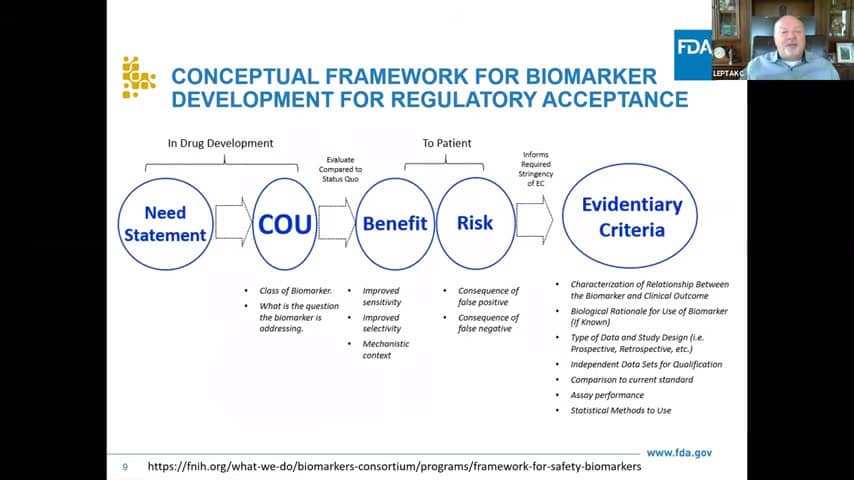
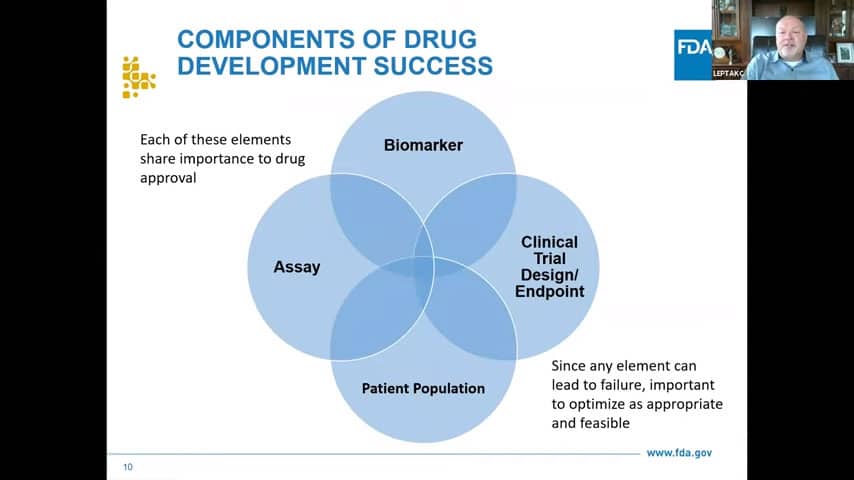
- Here are the elements of successful biomarker development for regulatory acceptance, taken from a paper that Allison mentioned, which we released so we can have better conversations about these elements to get you to your goal. We started with a need statement which for us is stated as drug development, which is sometimes confusing for folks trying to engage with us who are not trying to develop a drug themselves. Maybe they want to improve clinical care, which is a worthy goal, but not for our group, perhaps more for the devices group. So based on the Context of Use which we covered, we try to figure out what evidence we need to see based on COU and need, and we’re looking at benefits and risks for the patients, to help define the needed evidence. The evidence is going to include information around the analytics and the biomarker measurement, statistical analysis plan, in essence a fair bit of information that help support that biomarkers’ role.
- Simplified, but many times where we actually get success is when all those elements overlap. So when you’re starting a new process, we encourage defining what your assumptions are in the first place and actually clearly writing them down. Because if you get a negative result, unless you really do your due diligence up front, you don’t know what really caused the negative result. Clearly defining your assumptions will help you interpret your results better, so at the end you know what exactly might have gone wrong.
- Analytics and measurement, we define them broadly into two separate buckets – analytical and clinical validation. Analytical validation establishes that you can measure something, but doesn’t mean that it means anything. Clinical validation means that the measurement has biological clinical meaningfulness. That’s what we’re looking for in clinical data to help support whatever it’s COU might be.
- 21th Century Cures put us in a proactive role and gave us a much more collaborative process with a better structure, targets and timelines. This gives you a way to build a plan that you can then execute – Letter of Intent, Qualification Plan, and Full Qualification Package.
- Transparent decision making letters, shared learning culture. If you are developing an imaging biomarker, you can check a lot of information from other biomarkers and discussions around them, because you can see the past decisions. Over time we see a great improvement in the quality of submissions because of this even with people that we didn’t engage with before that much.
- You do not submit primary data for LOI, this is relatively short 10-20 pages tops, very focused and summary level information.
- Qualification is the bottleneck usually, it’s where we do a deep dive in science provided. Here’s where you’re saying what you see in science today, what’s the goal, what are the steps to get to the goal and how I intend to address the gaps. And if we agree on that, you can execute the plan knowing that if the data is supportive, we will be happy and won’t ask for more things down the road.
- Full qualification plan is where you submit all the primary data, analysis and interpretation and we do our analysis and see if we come to the same conclusion or not.
- Internally we have a three tiered review: First tier is within the program, we look at whether it is administratively complete, if you’re telling a clear story. What we find most of the people do is that they try to put too much information. We want to have a targeted conversation about a specific idea, so you need to be able to tell a clear story that is focused on a specific thing. Once you have a good story to tell we take our subject matter experts, look at it in earnest, put together recommendations and at last take it to a committee and the discipline chiefs.
- People are always interested in surrogates – there is a table of surrogate endpoints that is updated every 6 months.
- Finally if you are a partner or have an active IND with us, there are these new type C meetings for surrogate endpoints so that is something you can explore.
Q&A
Does the FDA do anything to improve biomarkers not submitted by outside applications? Is there a formal process to improve risk factors? Risk factors are important for balancing arms of clinical trials or doing stratified analysis post-hoc. What is the process by which science and medicine slowly make better versions of those? The example we are interested in this community is age, which is a risk factor for many diseases – this field has already produced a biomarker which is a better predictor for remaining years of life till death (methylation clocks) than chronological age. So it seems intuitive that at some point that should be used as a replacement for chronological age.
- We are interested in improvements, and usually it’s the owner of a related idea that drives that, so usually a pharma company. Part of a clinical trial is defining a risk factor, so the entity driving the trial is usually introducing the improvements as a part of the IND after a negotiation with the clinical division. Most of the biomarkers these days are composites, most of the time also with demographic and sociographic data, so age could be included as well. Maybe we’re not quite there with the biomarker you mentioned because for the biomarker to come in and be used as part of the risk stratification process, it would have to be done for a therapy that is targeting a disease that we recognize.
How do we get to the model where we use a biological age biomarker as a risk factor stratification model for example as something we balance the arms of a clinical trial with for COVID-19 instead of the number of years since birth?
- Present that as a part of the IND for a specific disease with age risk factor.
Does the FDA feel like it has a role of imposing a certain biomarker even if the clinical trial pharma sponsoring doesn’t necessarily have motivation to do that? Maybe it would make their drug look less efficacious if they had those true risk factors measured with best biomarkers available?
- I am not aware of any requirements that we can impose. We have tried to have better racial and ethnic representation, we encourage that with our sponsors, but we still have a very unbalanced white male clinical trial population. We can encourage but we can’t mandate.
What are the considerations/barriers for including a biomarker for X in a trial for Y? For example if there’s a biomarker for osteoarthritis risk factors, could that be included (as secondary or non-endpoint) in a trial where primary endpoint is cardiovascular events (in case one believes the drug could affect both)? Are there any sort of legal limitations there?
- Not that I am aware of, the current paradigm already allows this – you can target one disease and then look at expanded indications – that is pretty standard. I don’t know of a trial that is looking at 2 diseases simultaneously and has to be effective in both to be approved.
So in that case the main selection is probably patient selection, right? You have to have people who have those indications you are targeting and you need to be willing to pay for the measurements.
- Yes, it depends mainly on your resources. You know that for diabetes there is a correlation with cardiovascular risks – but it took a long time and long expensive trials for the companies to extract that claim and allow them to use it.
One interesting question for people in the aging community developing subtractive technologies is that the marker is the driver. So what you’re taking out (pathogenic target) is actually what you measure as a biomarker. So you are using an analytic technology to assess whether the therapeutic is depleting the target. Based on what you talked about today, it kinda looks like we don’t need to engage further in biomarker space other than the raw mechanistic confirmation that the target depletion analytic is accurate.
- I agree with that. Biomarkers do not need to be qualified to be used. There are diseases that have abnormal accumulation of protein. So there are therapies that were able to degrade that target and were approved based on that. So as long as what you are targeting has biological rationale to support its role in the disease of interest, then that should be sufficient if you can measure it and provide evidence about that.
I assume then we can just forward the validation data from the company doing analytics and that should be enough, right?
- Maybe yes, maybe no. In theory yes, but the data needs to be good, we need to see the data and how it was collected. If it is not up to our standards, we might need to see some other data.
A lot of the biomarkers we are having in the aging space are not necessarily the physiology, when we have epigenetic clocks, they might change when we treat aging but that doesn’t mean that they are the mechanism that we targeted. When it comes to aging we have to think a little bit differently then the silos and diseases we grew up with.
- I don’t disagree with that, there was a cluster sponsored at NIH a number of years ago that was interested in aging as well. There is an openness – we now have a handful of approvals in the oncology space, where the indication is not for a specific disease but a specific pathway shared by multiple oncologic tumors. So aging might be similar in that, maybe it is possible to target a pathway that affects multiple diseases and has multiple outcomes.
In your database of molecule based biomarkers, do you differentiate/record cell bound molecules vs soluble molecules?
- No. If it is a defining characteristic, it is a defining characteristic, whether it is free flowing or bound to something.
Are you seeing an increase in non-invasive imaging-based biomarkers, given progress in this area, especially for the brain (with more and more approaches based on light for example)?
- Early on during the start of the program like 15 years ago, it was mostly soluble markers that we studied. Now across our portfolio of 70+ programs, I would say at least half of them are imaging in some type or form. For some diseases even getting access to tissue and acquiring samples is costly or dangerous (like liver biopsy for example). So people are looking into doing something about that non-invasively. Around the brain people are looking at CT and MRI features for axonal shearing and other anatomic structures associated with cells. Even in psychiatry there are EEG signal programs helping differentiate between psychiatric diseases in homogenous diseases. If you’re looking at imaging just from a cost perspective, you also need to take into account the practicality for clinical trials and the specific disease. The likelihood that a pharma company will be doing PET scans for tens of thousands of people is pretty small.
In mouse models we have these miniature laser two photon cameras and the mice can run around, that can be much cheaper than these big MRIs, and they are light based, so you don’t have to penetrate the skull necessarily to get a lot of information. So anything like that yet for humans?
- Nothing comes to mind at the moment, maybe in devices, there are things. We are open to it.
A hypothetical question – imagine you started a company and you want to do something about aging. Right now, because aging is not recognized as a disease, you have to develop a drug against a certain recognized disease. And so you’re facing a kind of chicken and egg question, where you are forced to take these side routes through age-related indications, but they are not getting where they actually want to go. What would be your suggestions?
- I don’t know. Unless something changes around our statute, we are kind of bound to act within our confines. Obviously we’re approving drugs based on clinical benefit. So part of it is probably going to need to be some sort of societal lobbying saying that living longer is a benefit and therefore it should be recognized that way. At the moment the best approach is probably to focus on diseases that are part of aging and try to target those and try to expand indications into other age related diseases. That’s a stepwise approach within the paradigm that exists.
Cell senescence is known to be a hallmark of aging and underpin a multiple of age related diseases. Removal of senescent cells also improves health. If presence of senescent cells is known to contribute to a specific age- related disease and removal of senescent cells by senolytic therapies is associated with improved patient outcome (eg IPF) then would a marker of senolysis (eg Judy Campisi’s lipid – Cell Metabolism paper) be something that may meet criteria?
- If you are looking at senescent cells as a part of the causal pathway to a disease and you can say that if you impact those cells, there is a clinical benefit for that disease, I don’t see that as an unreasonable paradigm to have a conversation around. Many times we’re kind of targeting downstream effects, most of the diseases are going to be multifactorial, so I don’t see what you’re describing as that much different.
At what point might it be possible to determine it as a biomarker of aging rather than a biomarker of specific age related disease(s) after you expand it into multiple diseases?
- In terms of taking a marker of that cell and applying it to a different disease, we are always wondering what is the generalizability of a certain biomarker. Safety is an example of that. When we are looking at off-target effects and organ toxicity and they’re developing a set of novel safety biomarkers that should augment or replace our current ones, it’s being studied under a limited number of mechanisms of action. So then the question comes whether we will be able to generalize it more or just need to restrict it to that set of mechanisms of actions. It always goes down to the understanding of the underpinnings of the biology being used. So if you can say that this particular senescent cell is integral to multiple different diseases, and you have a biology biomarker for that cell, initially you might want to target one or two, and from there you can get to a generalized point beyond that.
With the type C meeting, do you get involved in it too, or does the division handle it on it’s own? At what point do you get involved?
- Request goes to the division, but it’s our group that is in charge of the meetings and that created the background materials for it and goes through with it. We work with the division and then take it to the committee, which acts as an advisory and has supervisory inputs.
On the subject of imaging-based biomarkers. There are types of data that are very easy to collect, such as facial headshots and recordings of voice, that can be used to create useful biomarkers for age, neurological disorders, etc. Given that this data can be collected trivially (and machine learning can be used to make sure such data collection is normalized easily) – what would it take to have the collection of such data become a mandated collection component of *any* trial – so that this data could always be there to later look at secondary indications like effect on aging.
- I don’t know of any mechanism for FDA-wide requirements or mandates. Pharma companies are certainly not going to be happy with the addition of new data collection given the expenses on clinical trials are huge already.
In that case what’s the right pathway or leverage to push? How can we advocate for it?
- I don’t really know. But we are looking at these things, for example for eye tracking for autism spectrum disorder, and that is also a biomarker that we are looking into in Alzheimer. The openness is there, but the hurdle is probably on the other end – which diseases are we going to target and how to convince the pharma partners. Focus on the people who have the power, and for whom these problems are in their paygrade. Commissioners, secretaries, folks in congress,…
Seminar summary by Bolek Kerous.