Summary
When attempting to manipulate cellular function, it’s much easier to use the established regulatory pathways rather than building your own. Inserting photoswitches at key points in those pathways let us control cell function with light. The best location for such switches is on the membrane of the cell – easy to measure, open to light sources, and where most of the cell’s regulatory proteins are located.
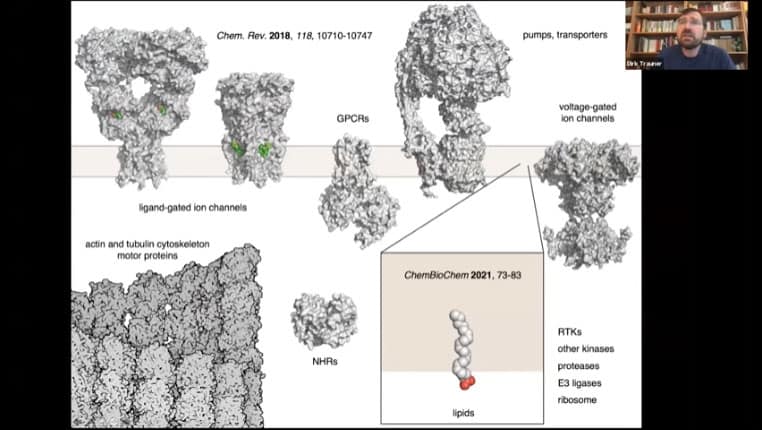
Voltage gated ion channels, ligand gated ion channels, pumps, actin and tubulin cytoskeleton motor proteins, nuclear hormone receptors, lipids, E3 ligases, and g-protein coupled receptors have all been successfully married to photoswitches so far.
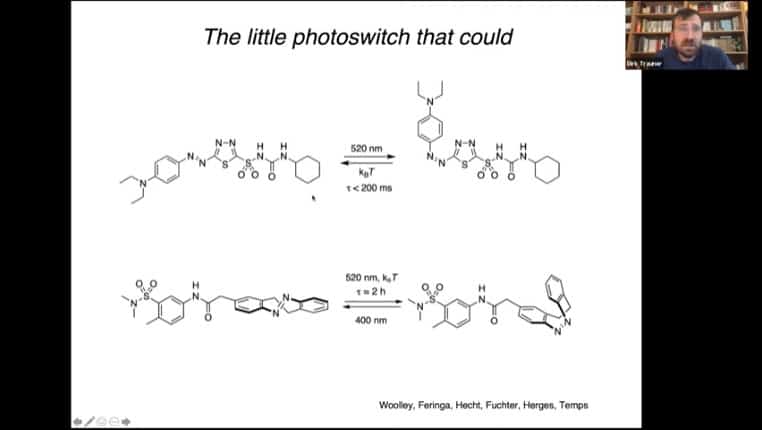
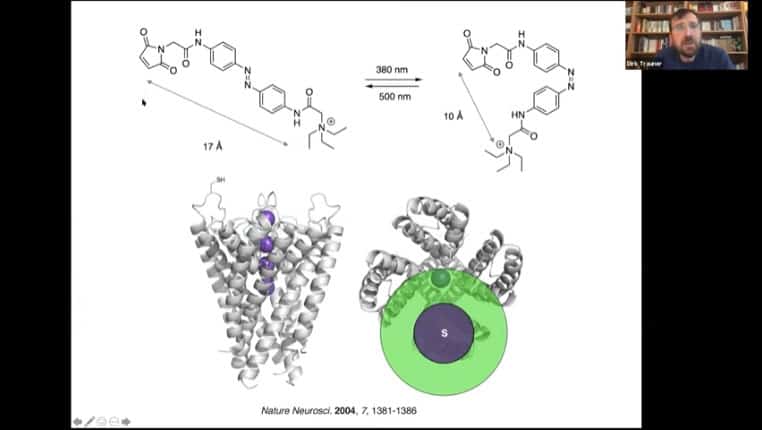
A key molecule is azobenzene, which switches conformation based on exposure to either 500nm or 360nm light.
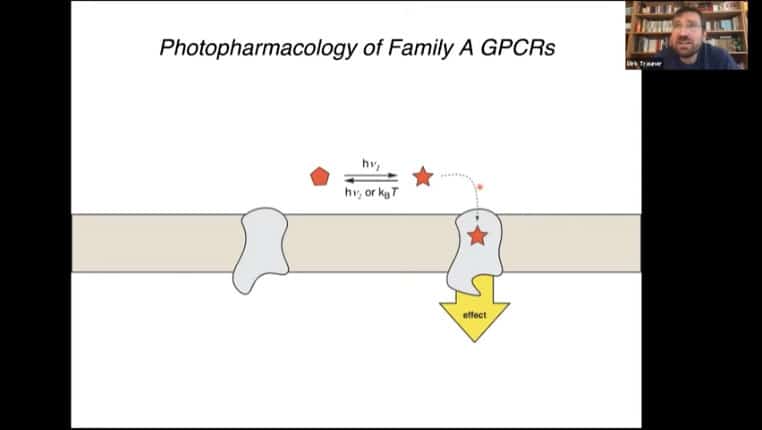
Tethering ligands to receptors using azobenzene molecules in the tether allows you to directly control bioactivity across a wide spectrum of enzymes. Rapid controlled initiation of downstream signaling can be accomplished with the flick of a light switch.
Opportunities
Medium term goals – the possibility to control things with input signals other than light. Using magnetic fields or ultrasound would be more penetrative. It should also be possible to put molecular motors on proteins.
Presenters

Dirk Trauner, NYU
Dirk Trauner was born and raised in Linz, Austria, studied biology and chemistry at the University of Vienna, and received his undergraduate degree in chemistry from the Free University, Berlin. He then pursued graduate studies in chemistry under the direction of Prof. Johann Mulzer, with whom he moved to the University of Frankfurt and then…
Presentation: Controlling the molecular machinery of life with photoswitches
- Examples of proteins that have been modified over the years that have been modified with photoswitches
- Azobenzene is a molecule that responds to light by changing its conformation, and can be modified to alter its functionality
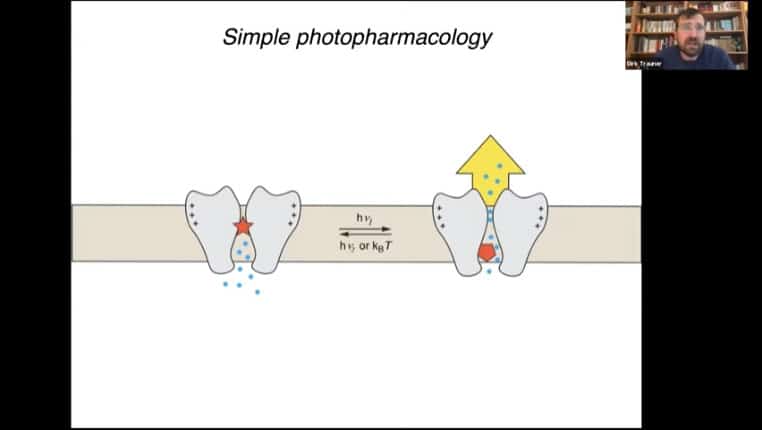
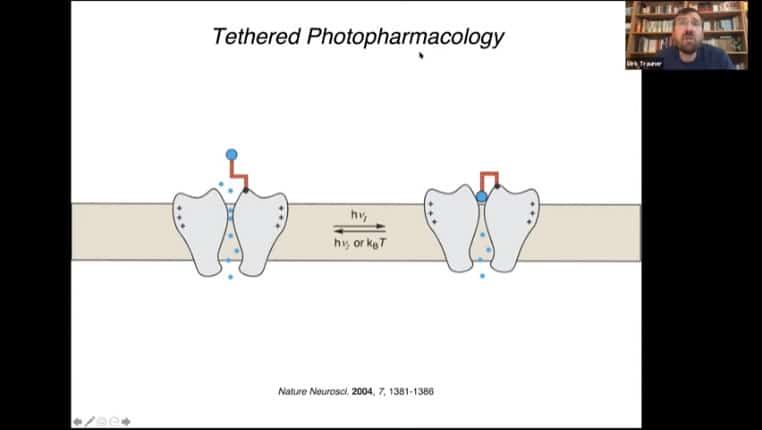
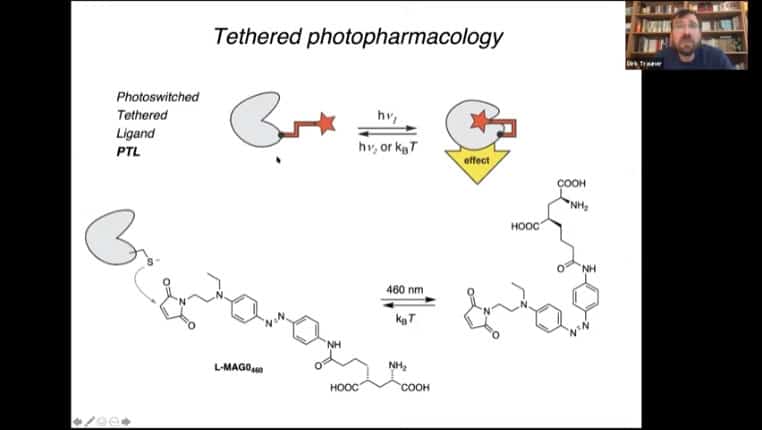
Example of a simple photoswitch that blocks an ion channel, and a tethered version of that photoswitch that uses a binding protein attached to a photoreceptive tether to alter protein function.
Diagram of tether mechanism
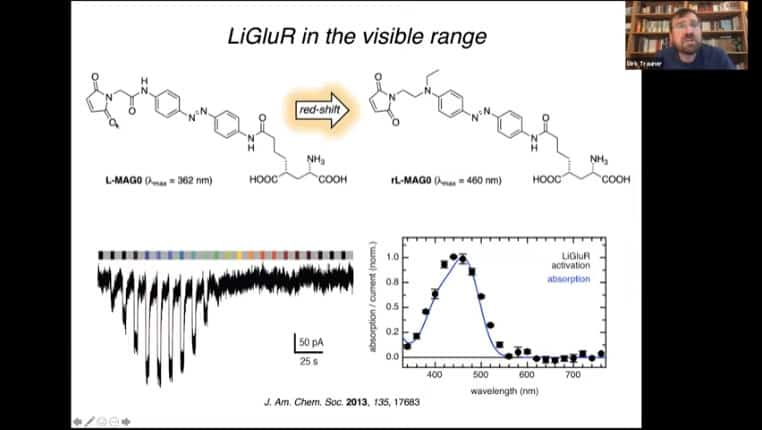
Alteration of the tether mechanism to respond to different light
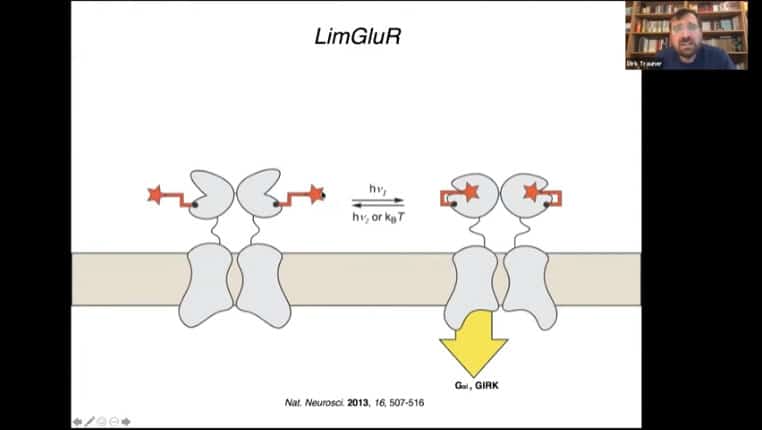
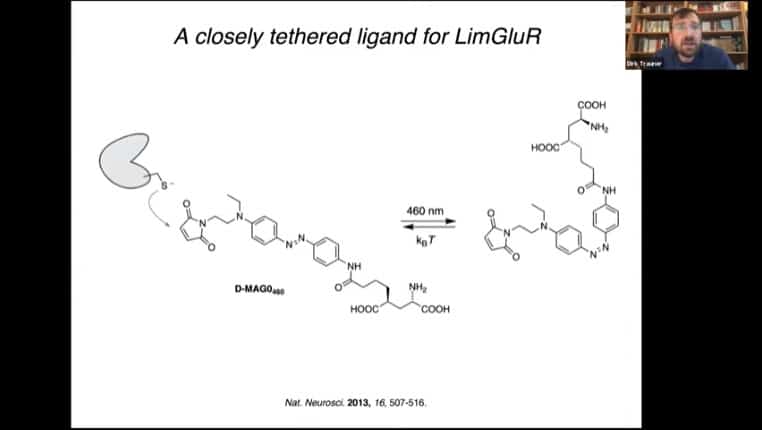
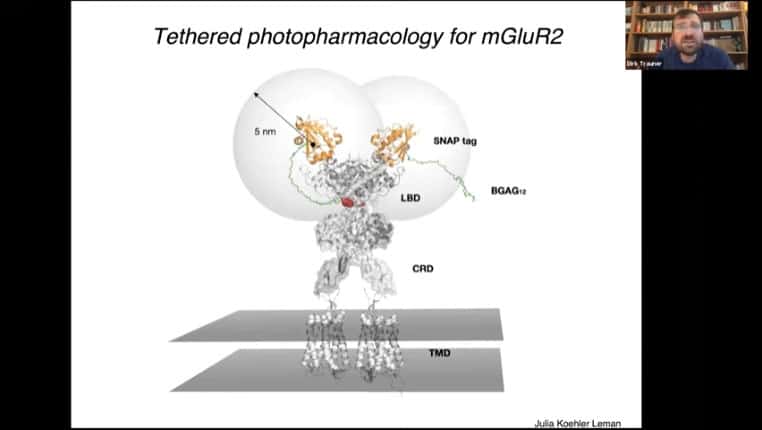
Tethering mechanism for a glutamate receptor
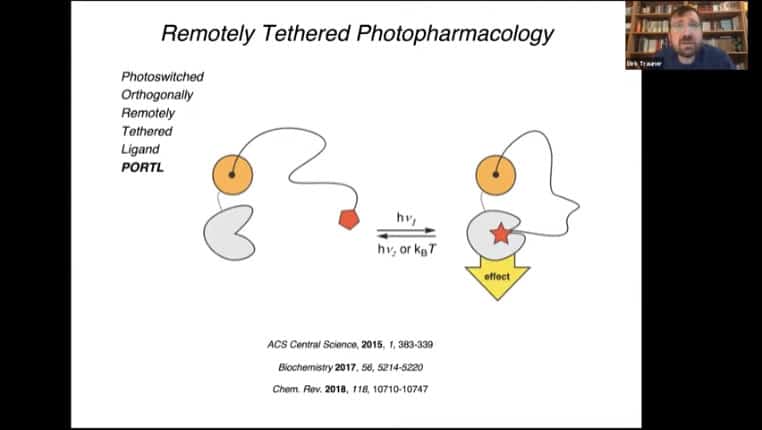
PORTL – Photoswitched Orthagonally Remotely Tethered Ligand, this mechanism acts like a dog on a leash where the ligand can move around in a radius around the receptor. Light alters the tether and causes activation of the receptor.
Another set of diagrams for PORTL versions of glutamate receptor tethers.
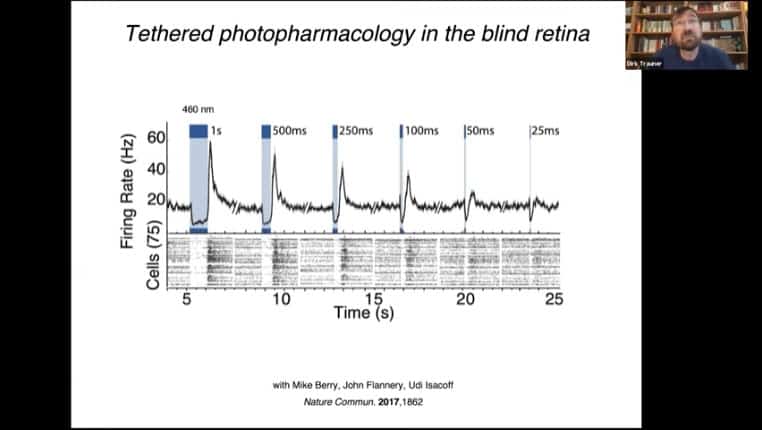
These mechanisms can be used to treat blindness.
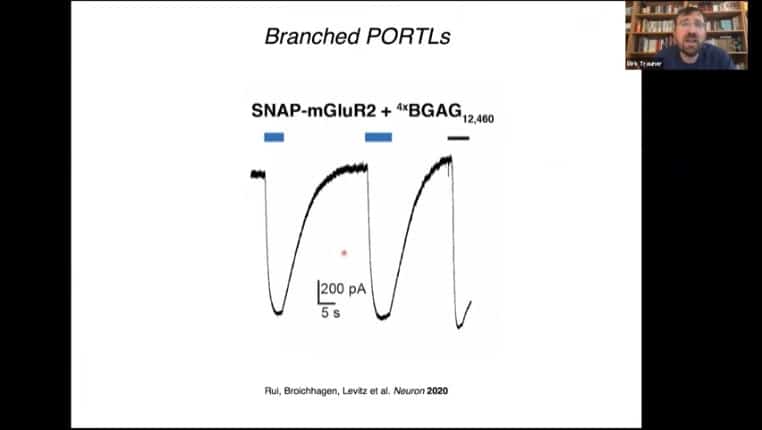
Branched tethered PORTL’s can increase the efficiency of the light activation mechanism.
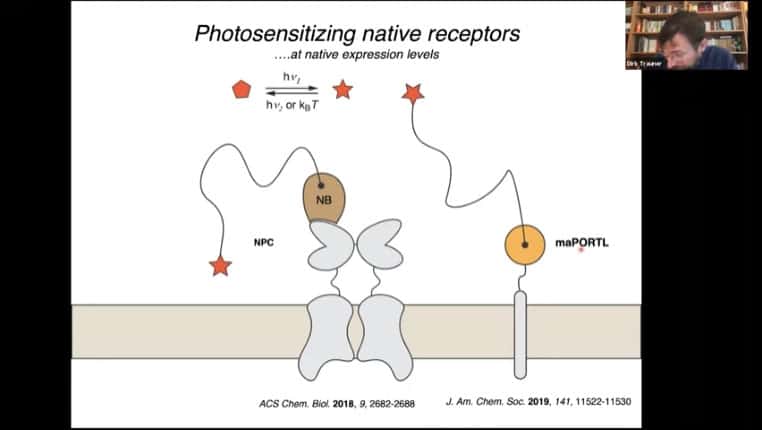
Nearby membrane bound proteins with photosensitive ligands can be used to increase the efficiency even further.
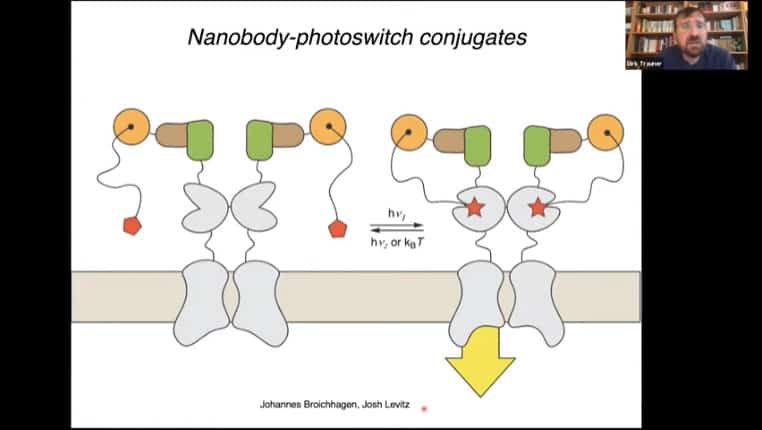
Nanobody photoswitch conjugates
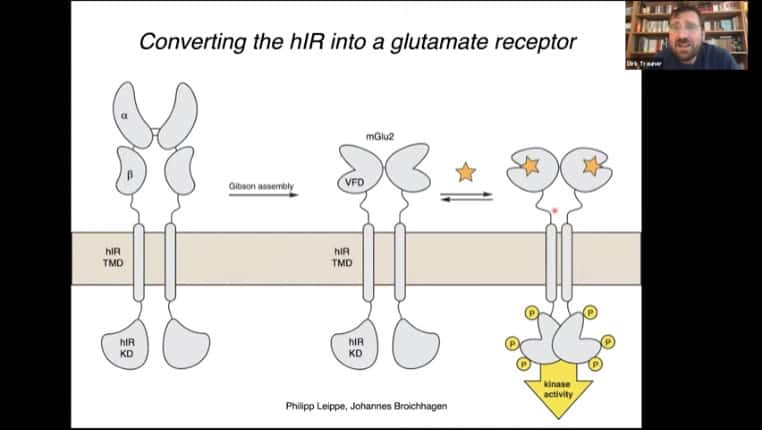
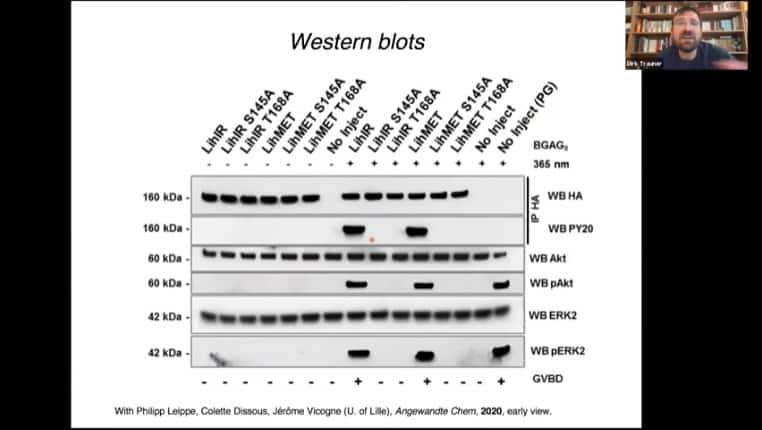
The insulin receptor can be converted into a glutamate receptor, then modified with the photoreceptor domain. You can then activate the insulin receptor pathway using light.
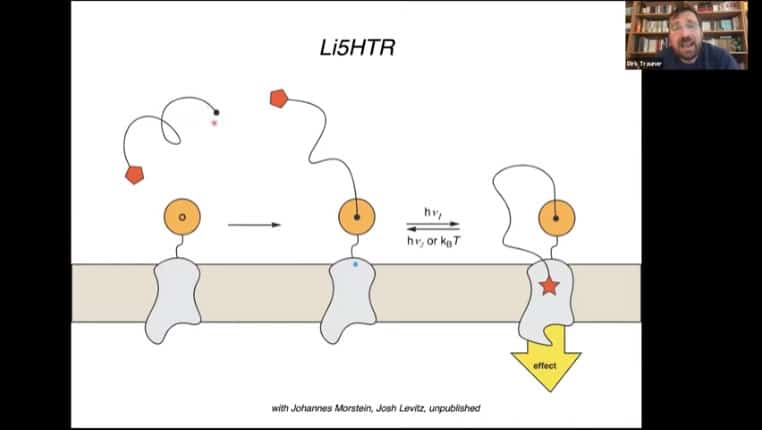
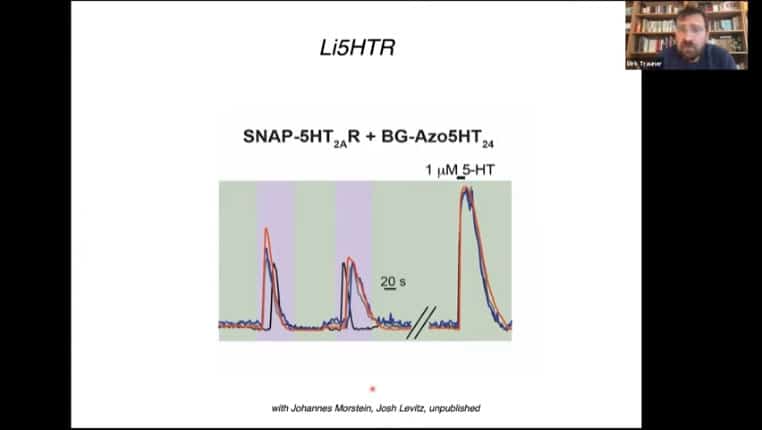
The 5HT receptor can also be modified with the photoreceptor mechanism.
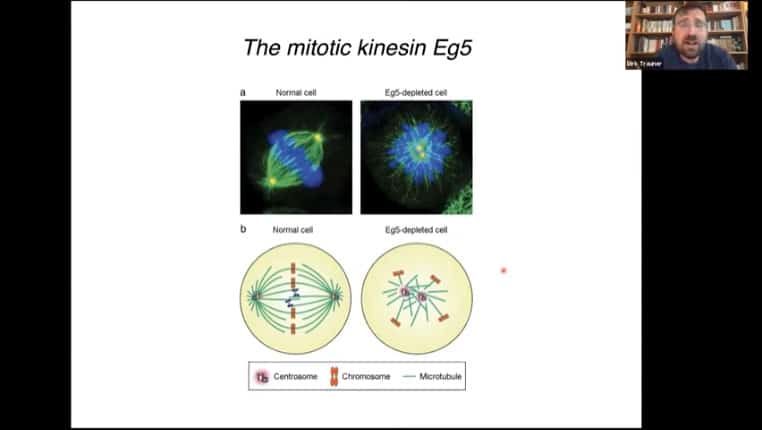
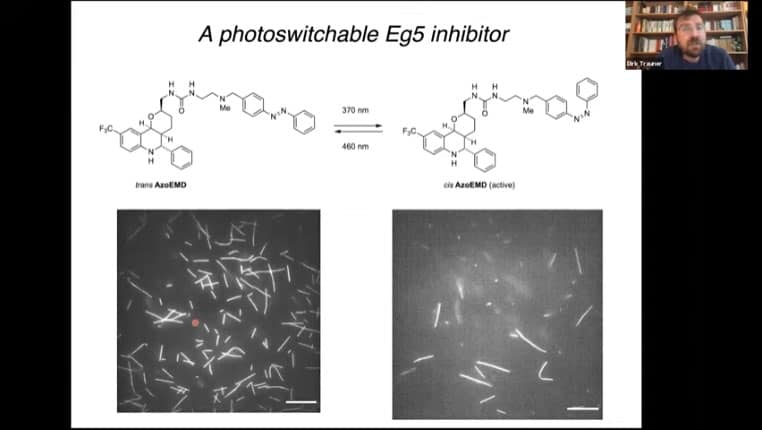
The kinesin Eg5 coordinates the polar functionality of mitotic spindles. It is possible to generate an Eg5 inhibitor that contains a photosensitive switch.
Many other lifecycle proteins have been altered to be photoswitchable as well.
Seminar summary by Aaron King.