Summary
Yunyan Qiu and Liang Feng describe the mechanism and possible uses of nanoscale molecular pumps. These pumps are constructed to attract and concentrate rotaxanes. Reduction and oxidation (redox) reactions drive the pumps, changing the polarity of the rotaxanes such that they can be driven across the pump cassette and onto a strand.
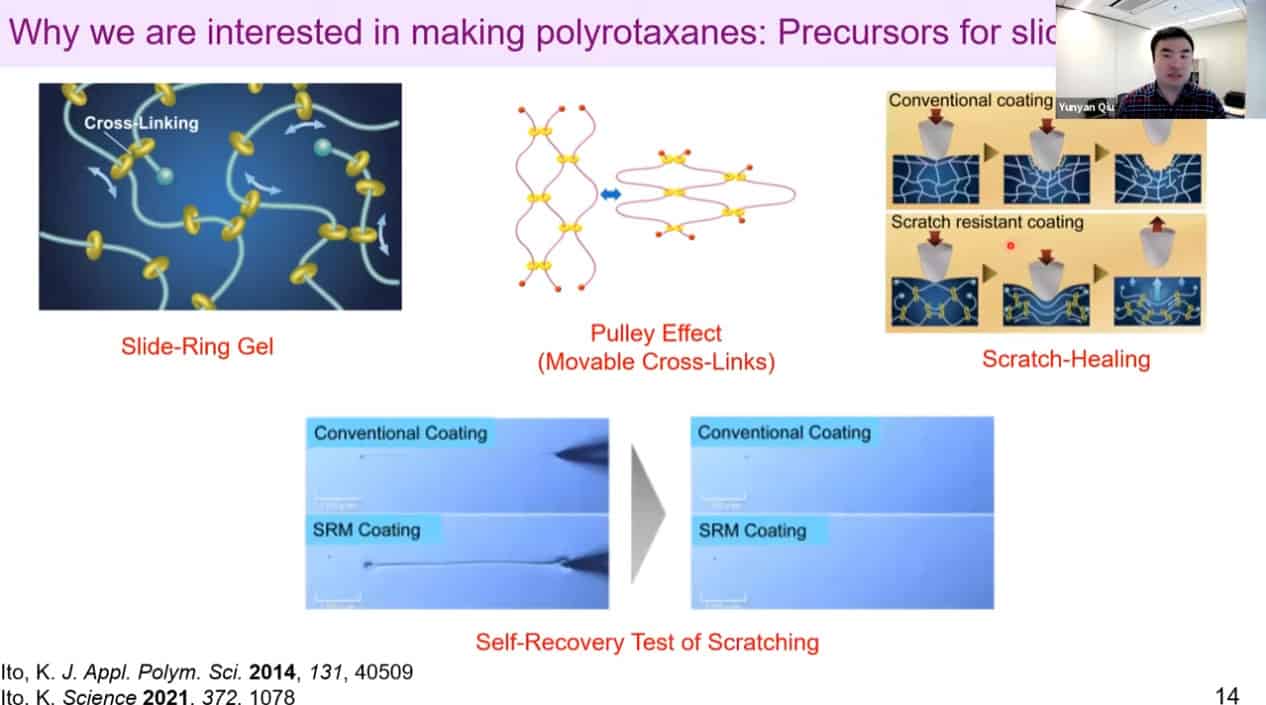
Some potential applications of this technology include power storage and sequence-specific polymer construction. Polyrotaxane threads can be bound together to create a mesh, which may lead to scratch resistant or self-healing materials.
These molecular pumps can work in suspension, but there may be advantages to mounting them onto a surface.
Validation of the molecular pump operation is done using nuclear magnetic resonance or mass spectrometry.
Presenters

Yunyan Qiu, Northwestern University
Yunyan Qiu is currently a postdoctoral fellow with Sir Fraser Stoddart at Northwestern University (NU), where he researches in the field of molecular machines and related functional materials. Most of his work at NU is to design and harness artificial molecular machines (AMMs) to produce functional polymers and materials in a…

Liang Feng, Northwestern University
Liang Feng is currently a postdoctoral fellow at Prof. Fraser Stoddart’s group at Northwestern University. He earned his B.Sc. in Chemistry from Wuhan University in 2016 under the guidance of Prof. Hexiang Deng, and obtained his Ph.D. in 2020 from Texas A&M University under the supervision of Prof. Hong-Cai Zhou. During this graduate…
Presentation: Artificial molecular pumps from solutions to surfaces
Yunyan Qiu
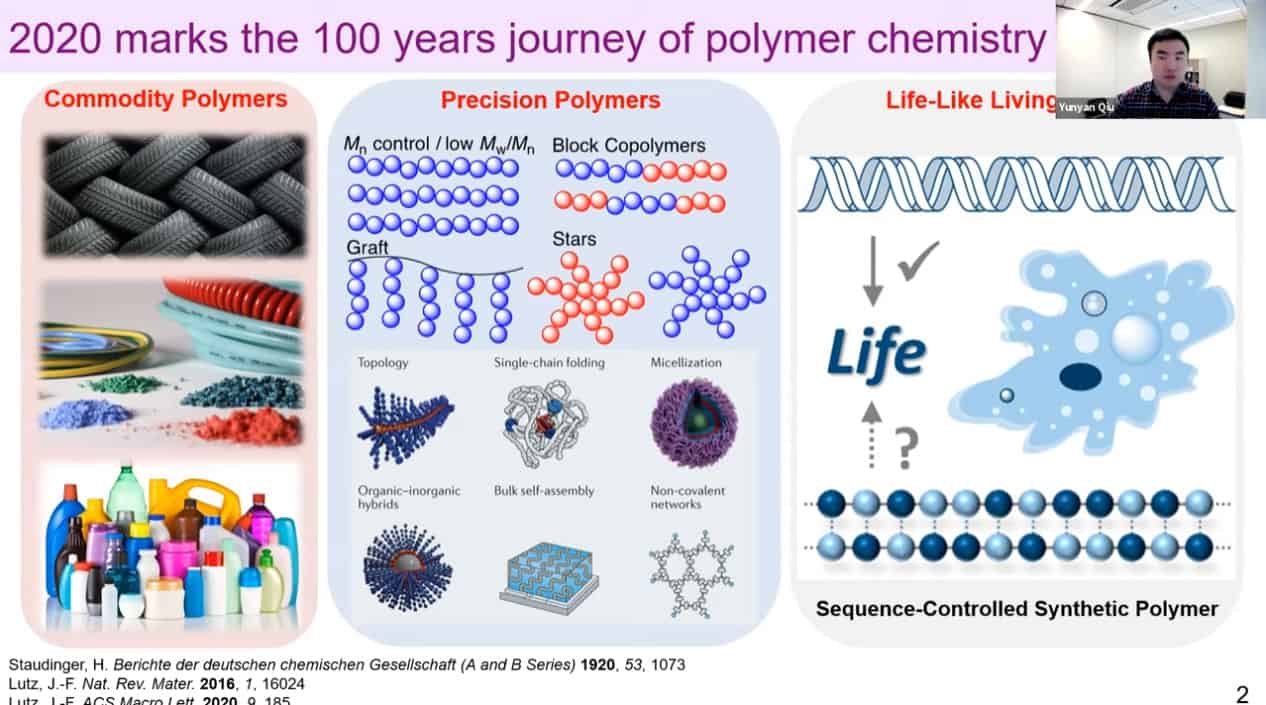
- 2020 marks the 100th anniversary of polymer chemistry. Commodity polymers have revolutionized society. Precision polymers have progressed significantly over the last 30 years. Natures polymers are the next step for polymer synthesis.
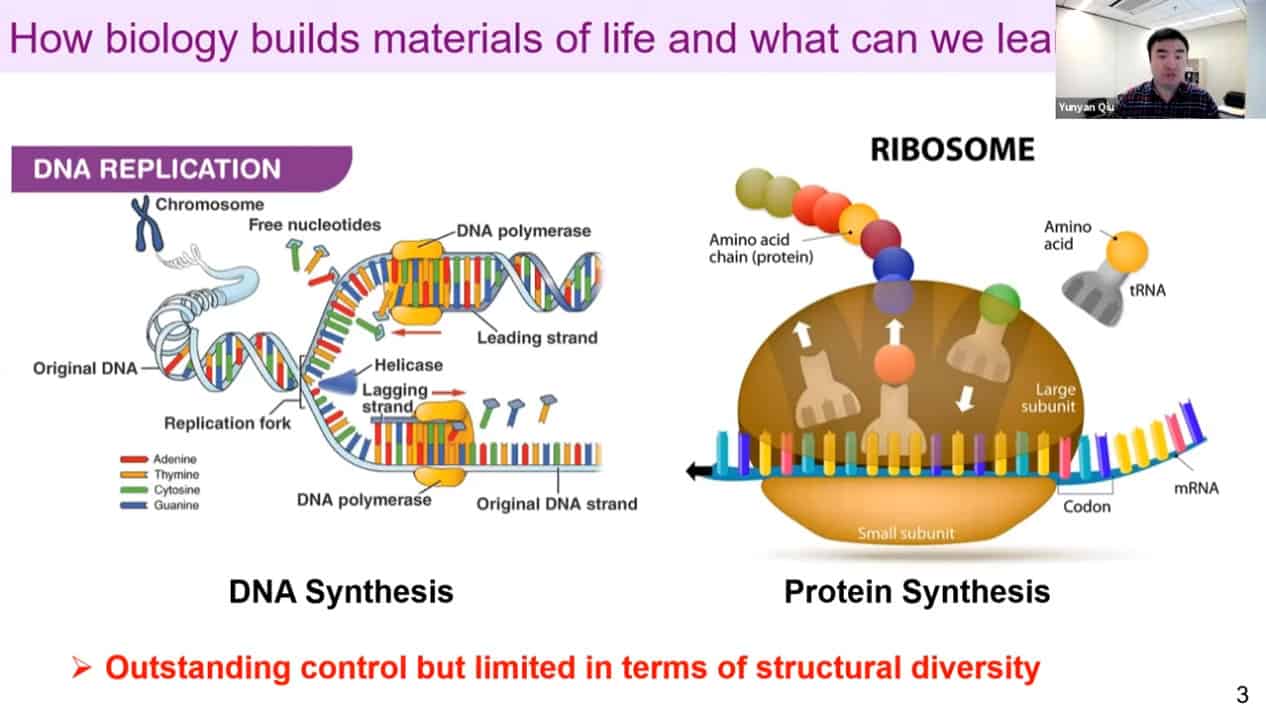
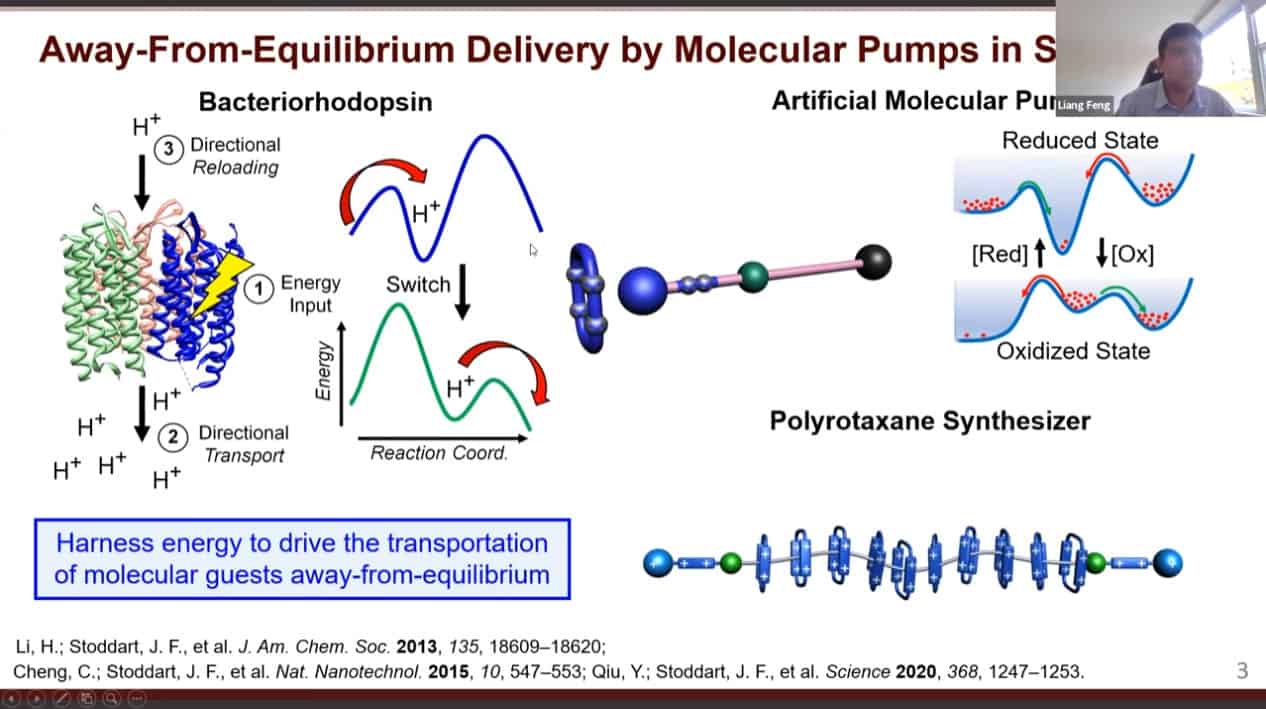
Nature uses biomolecular machines to build the materials of life. These machines have great control but are limited in diversity.
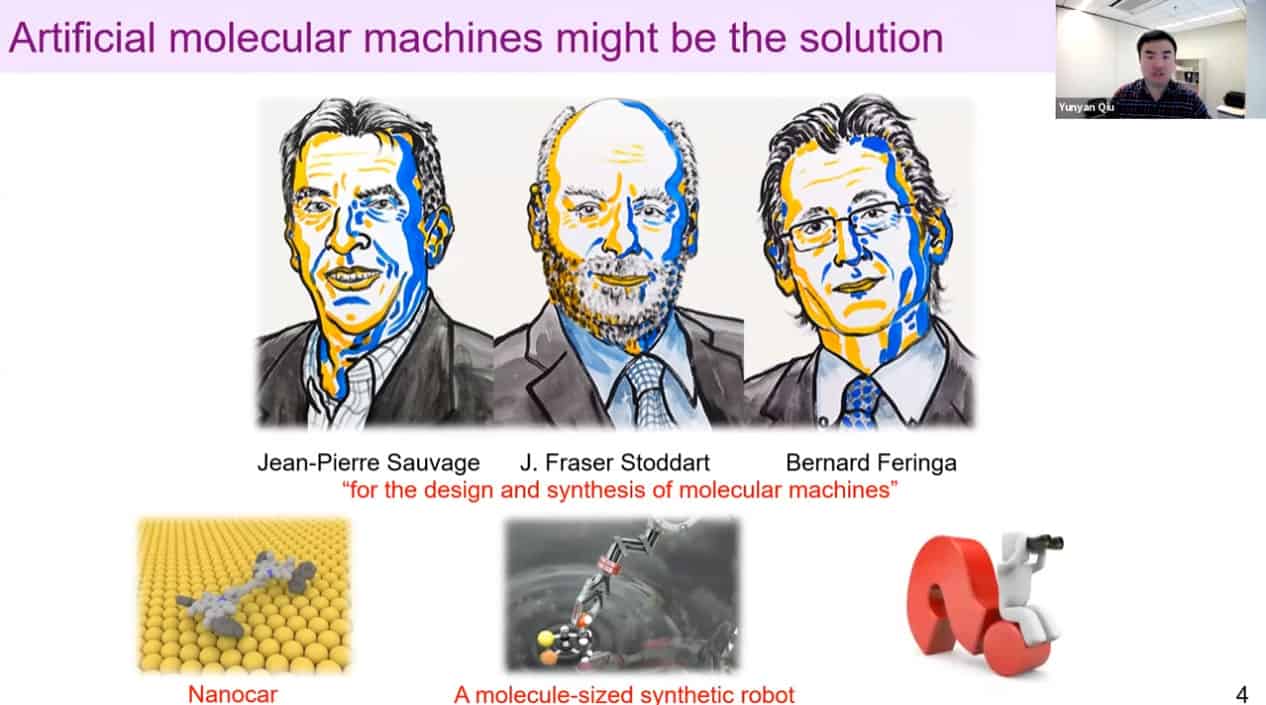
Nanocars and molecular robots have been created – but what is the next goal for artificial molecular machines?
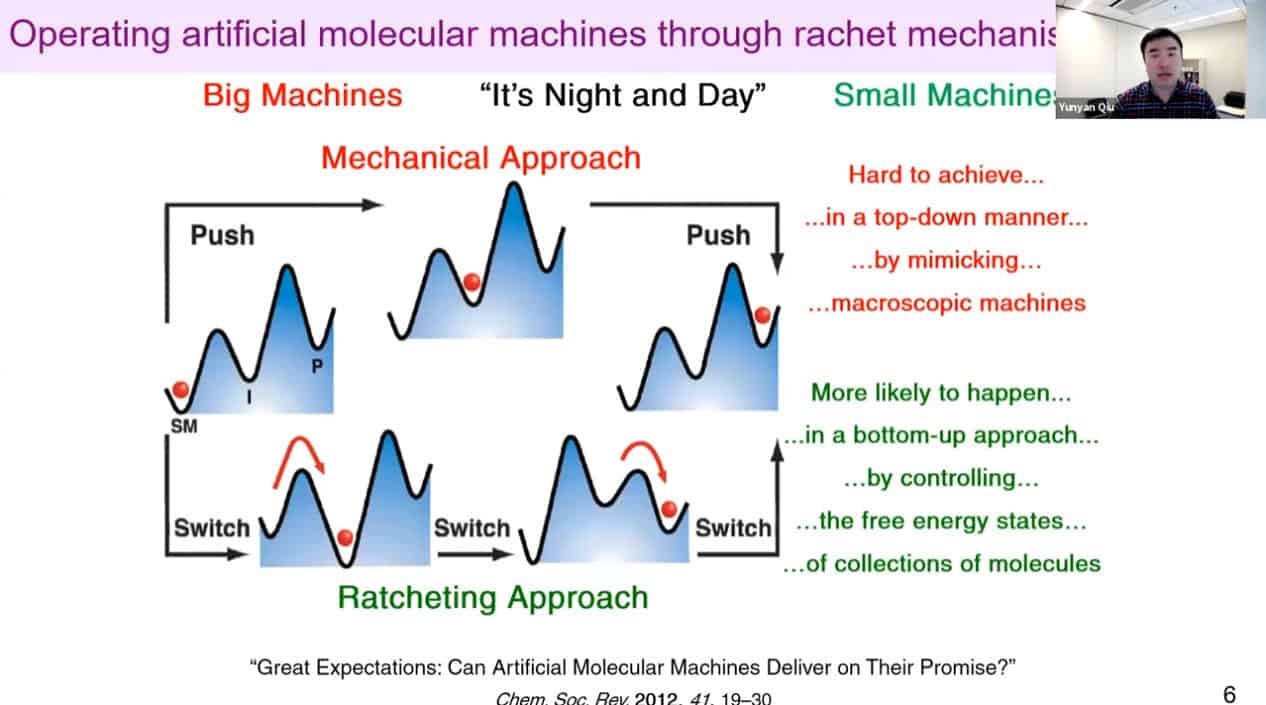
“Swim in molasses, walking in a hurricane” the movement at different scales behaves differently.
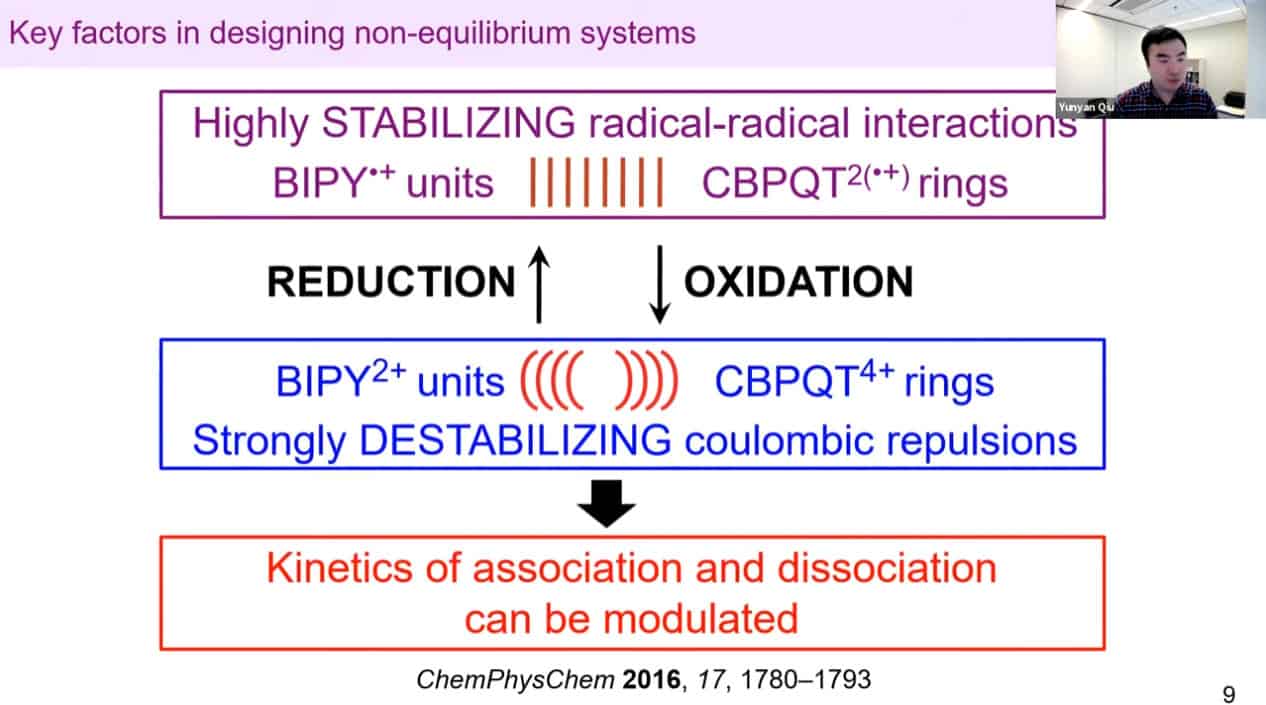
A ratcheting approach to movement vs. a mechanical approach.
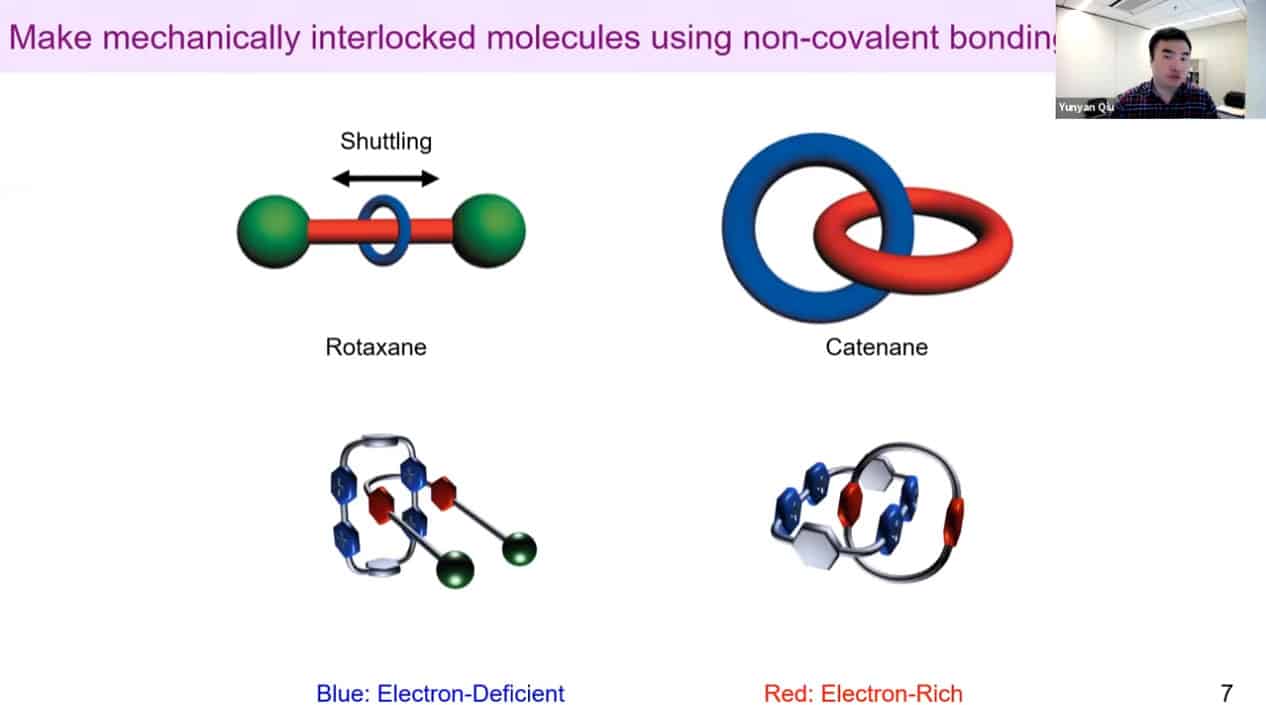
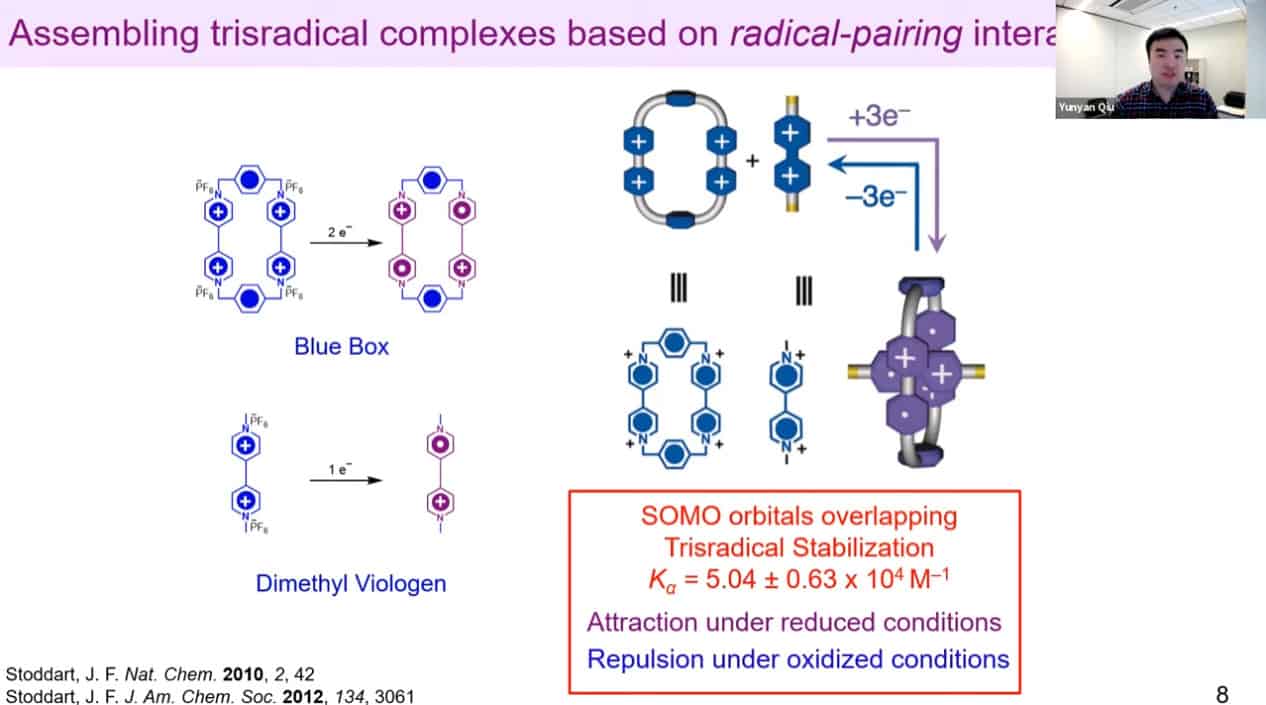
We need to develop molecules that can trigger self-binding via noncovalent interactions with each other – rotaxanes and catenanes work well for this.
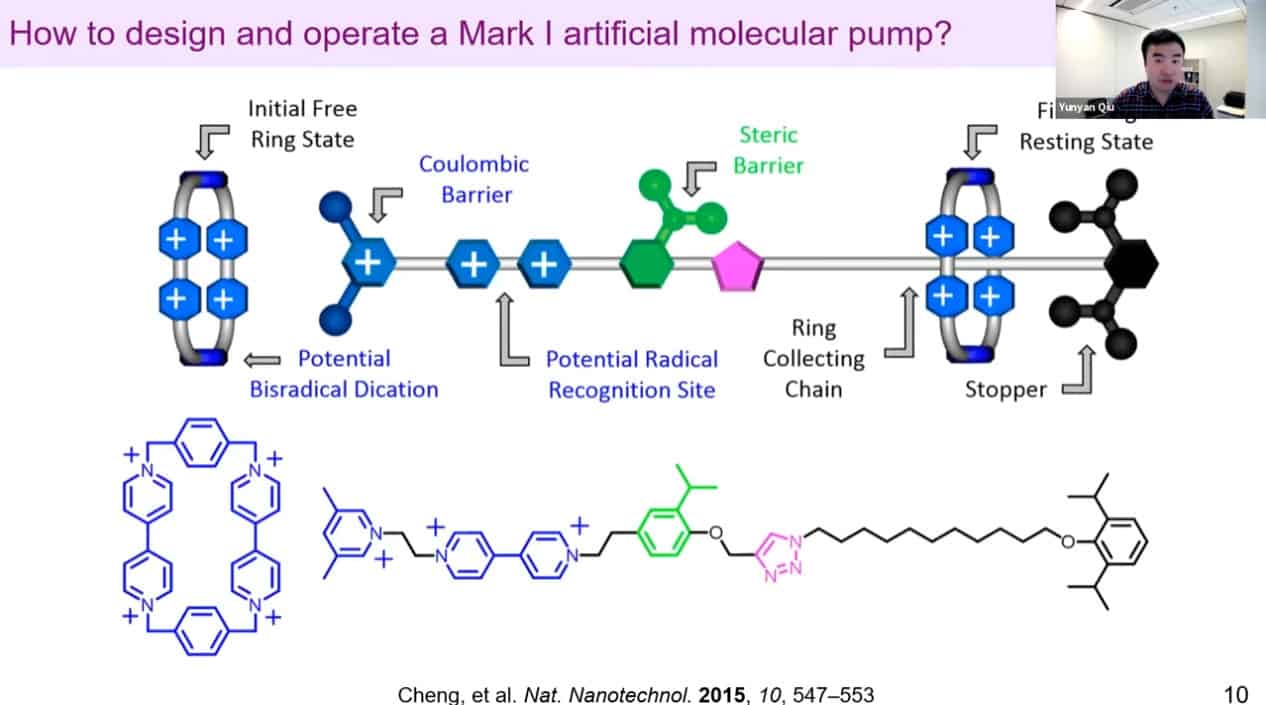
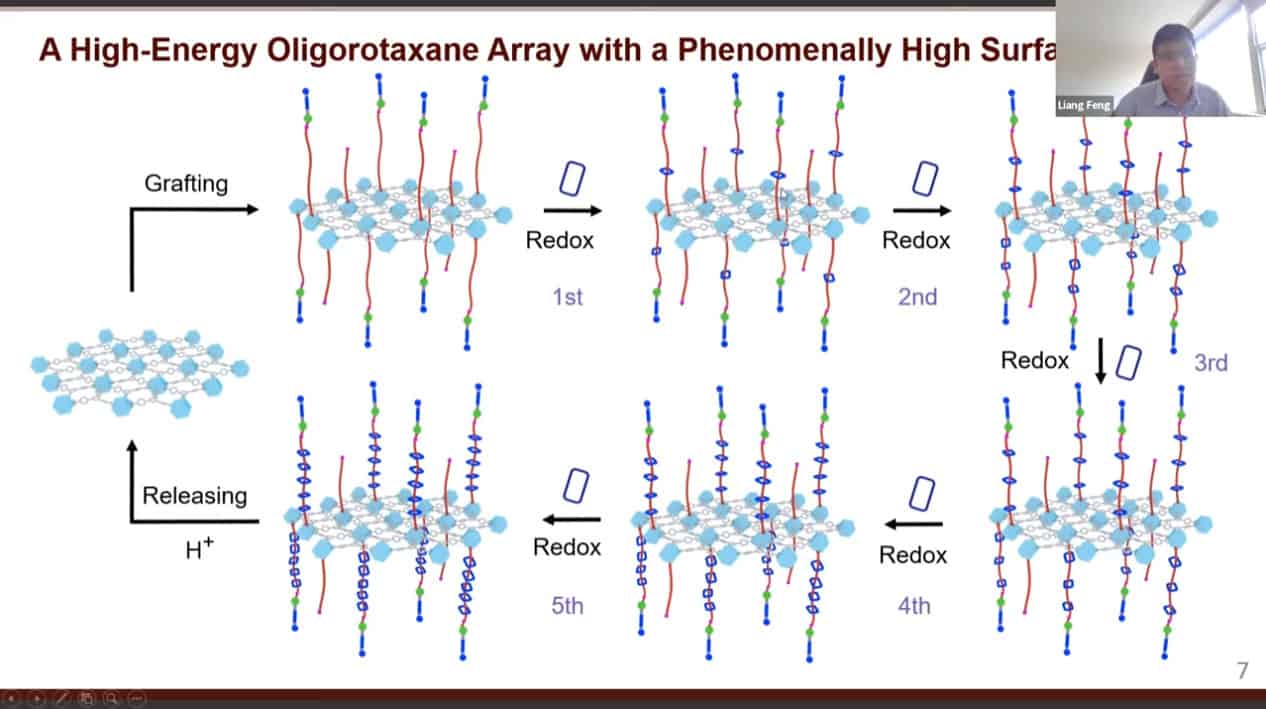
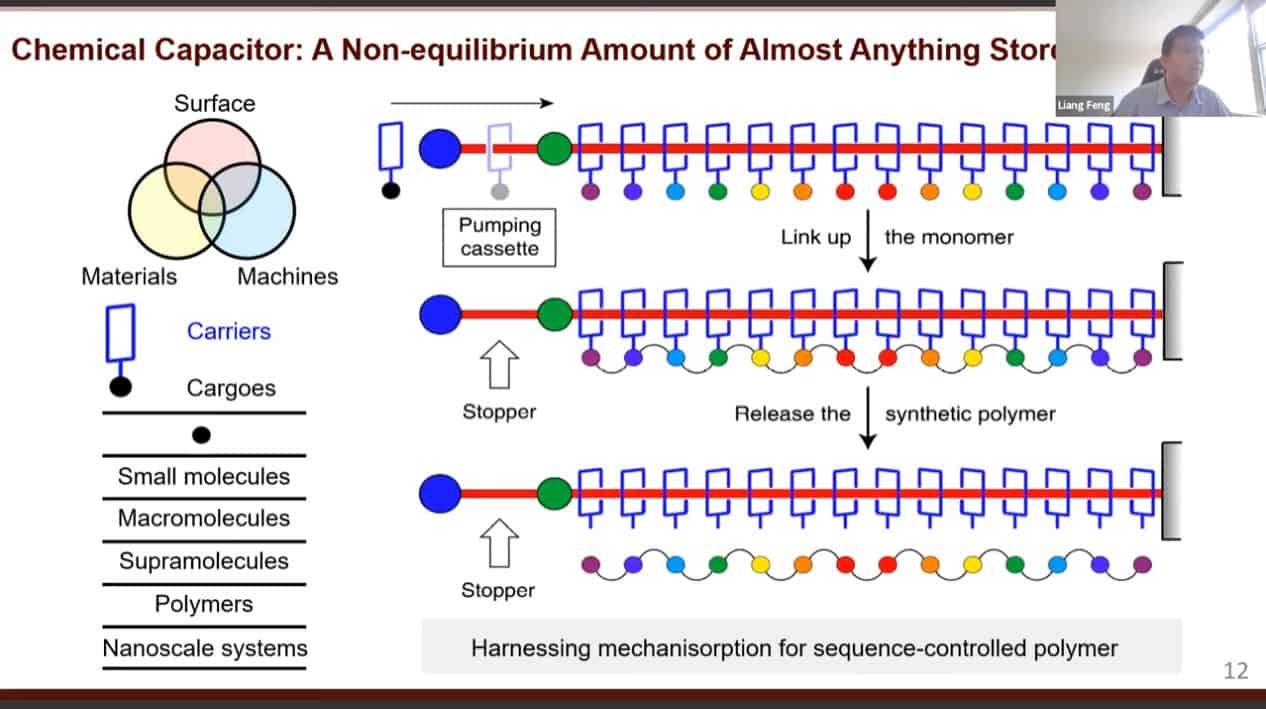
A design of an artificial molecular pump.
The mechanism of action of the artificial molecular pump.
Molecule position can be measured and shown to move away from equilibrium. The design of this pump has been improved by altering its structure slightly.
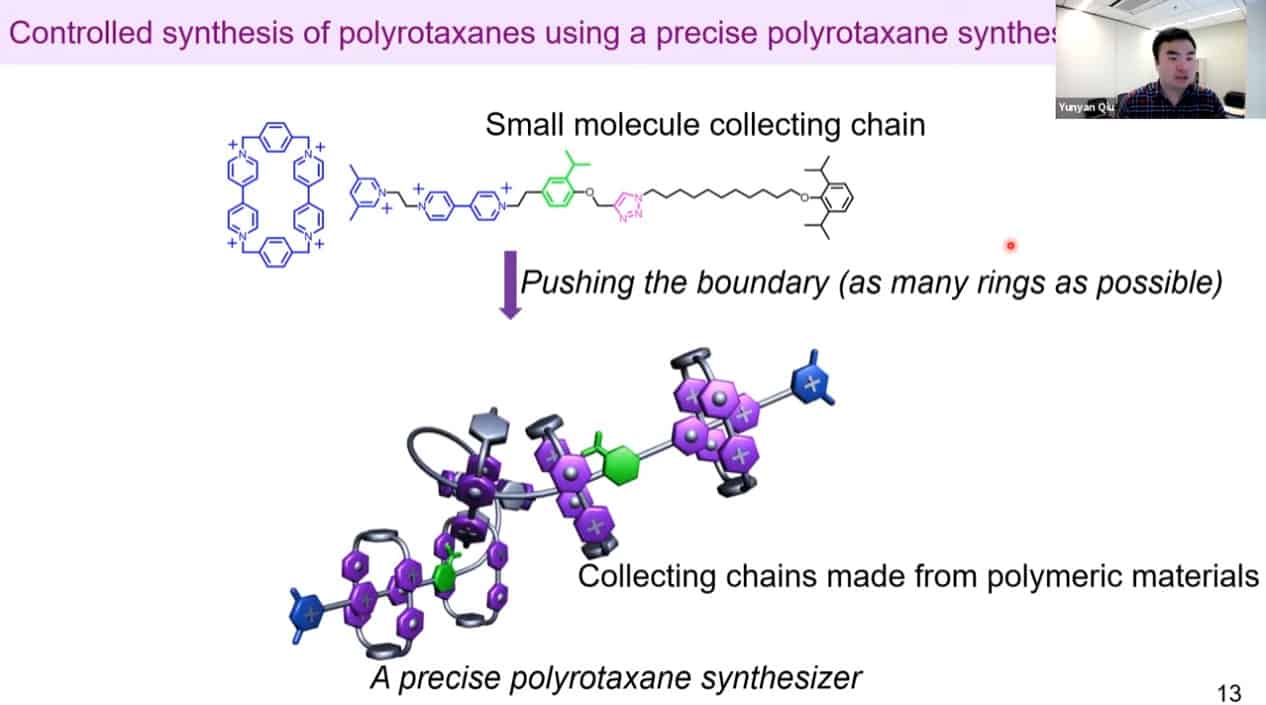
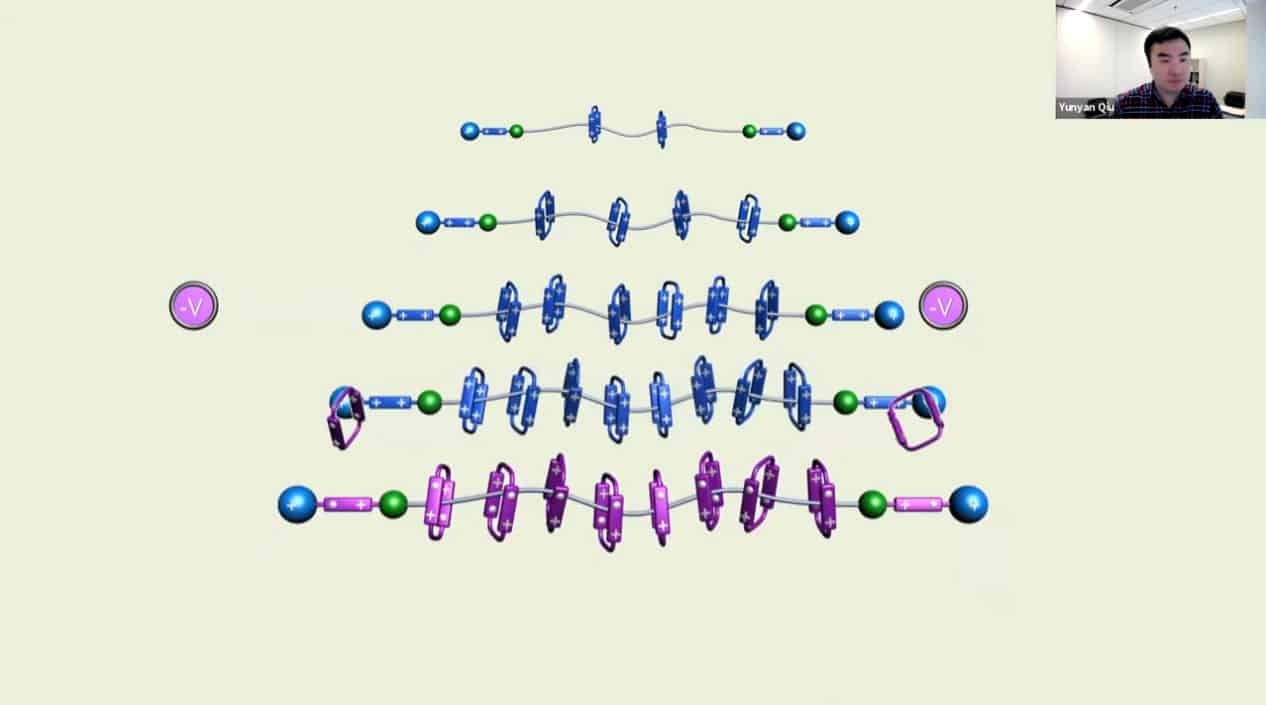
Multiple rotaxanes can be threaded onto a single pump.
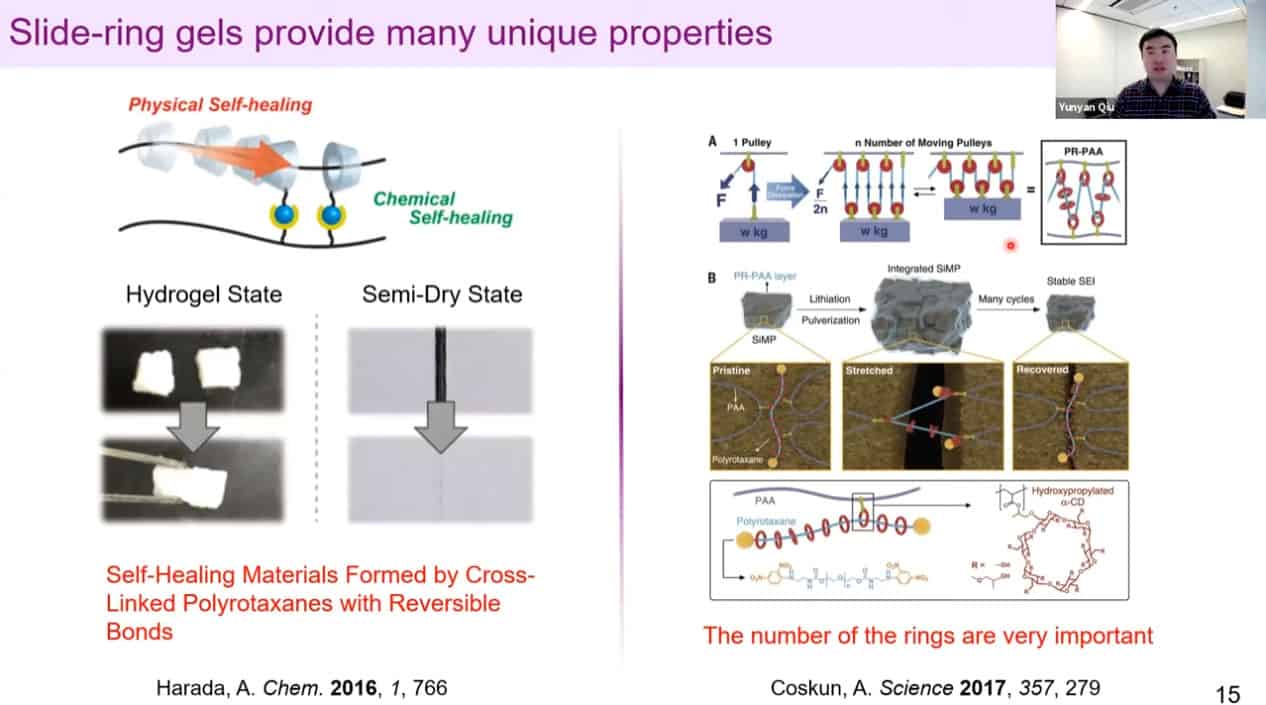
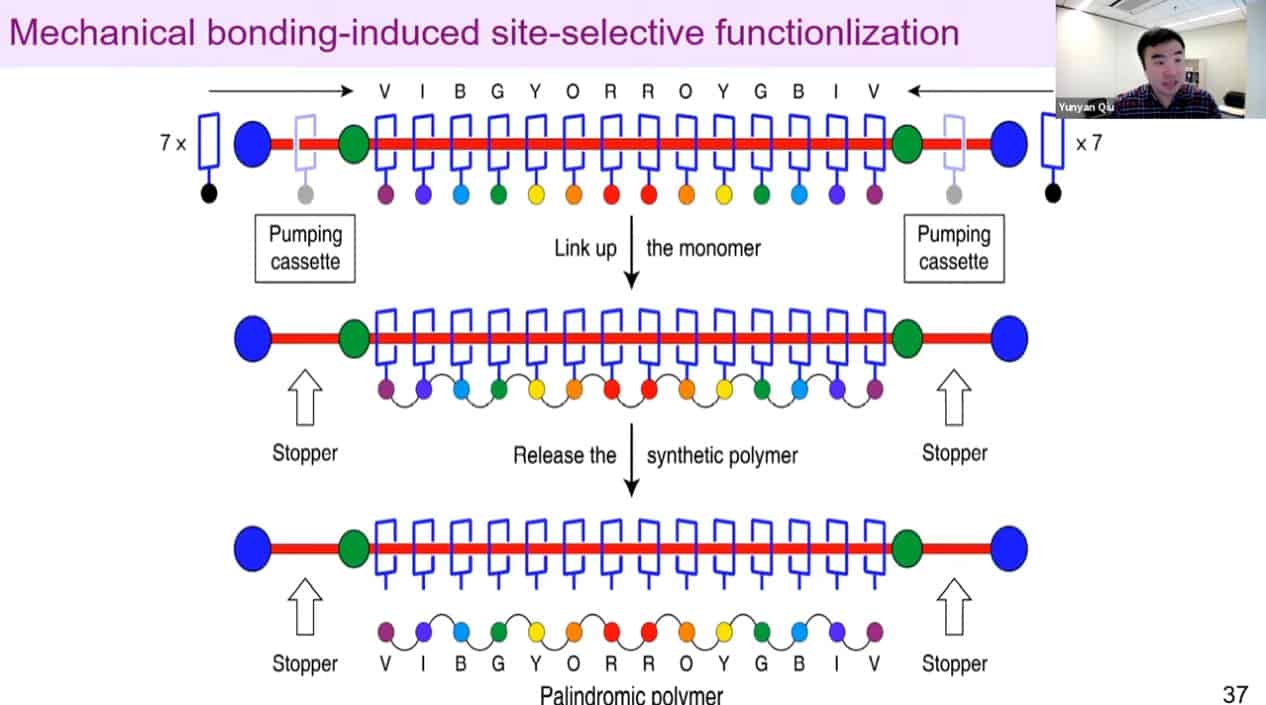
Polyrotaxanes can be linked together in a network to form self-healing coatings.
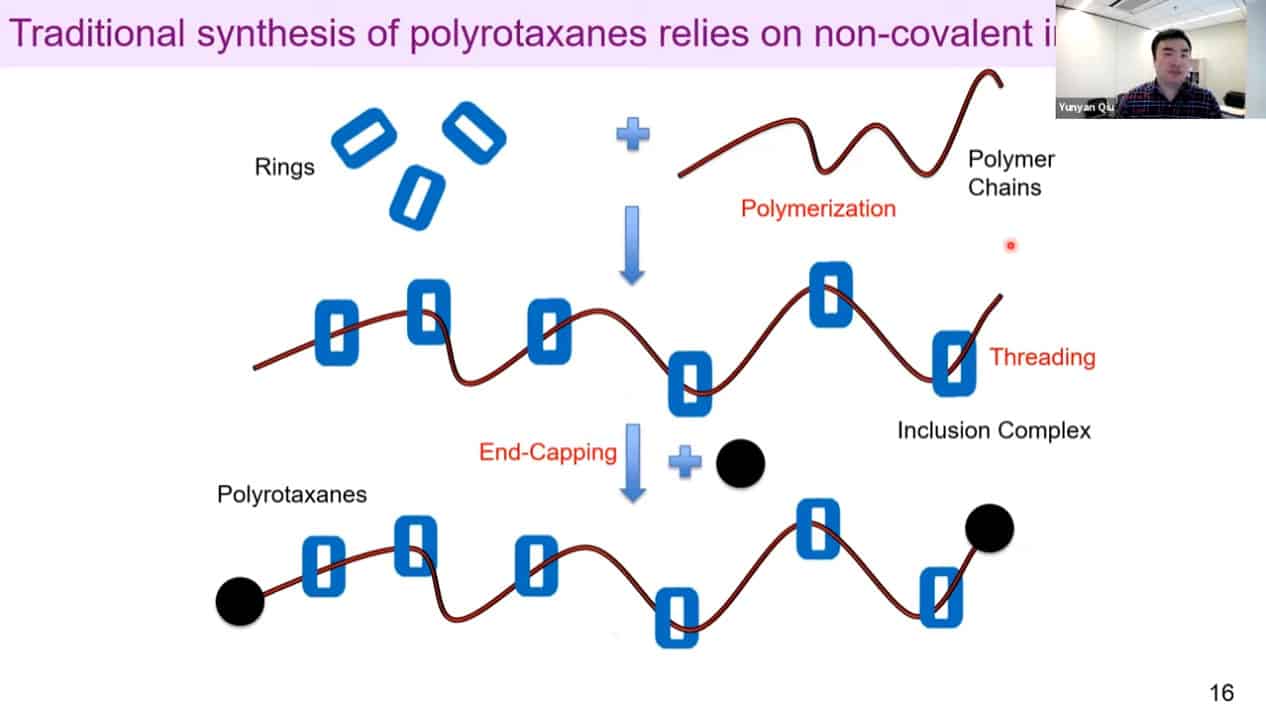
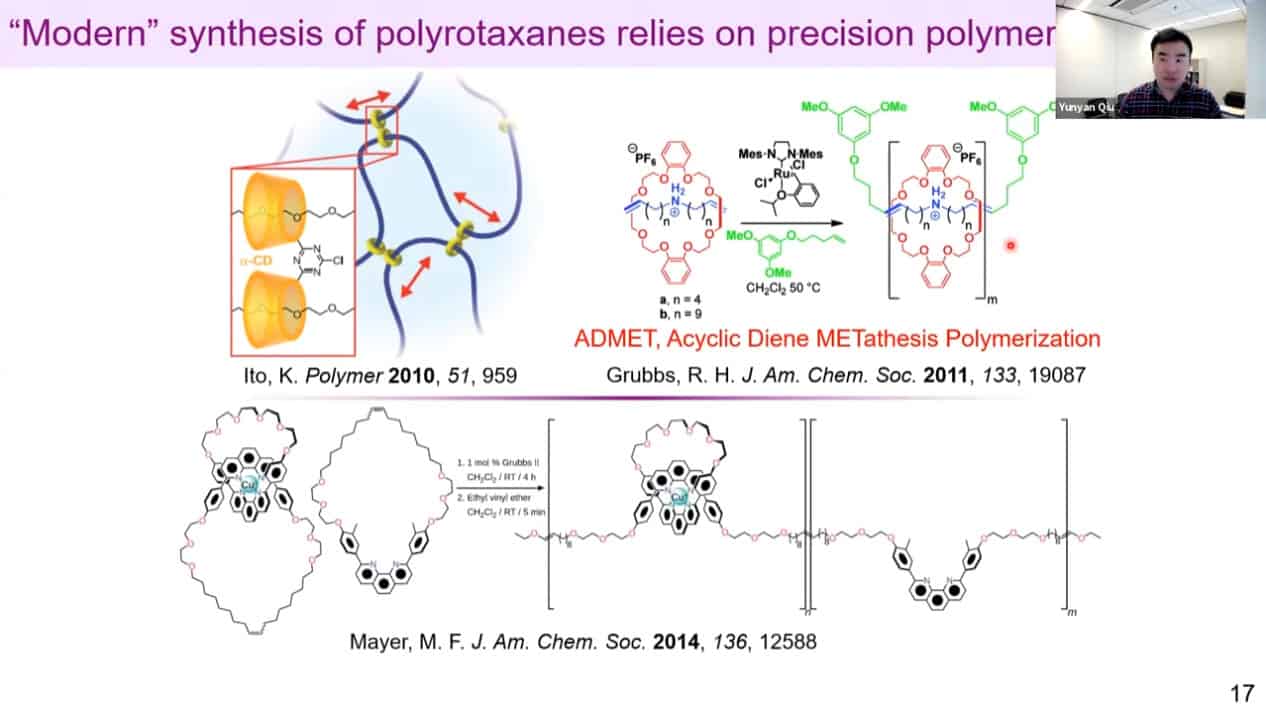
Traditional synthesis relies on non-covalent interactions and has limited control over rotaxane density.
A more advanced version uses molecular gates to control density, but there is still room for improvement.
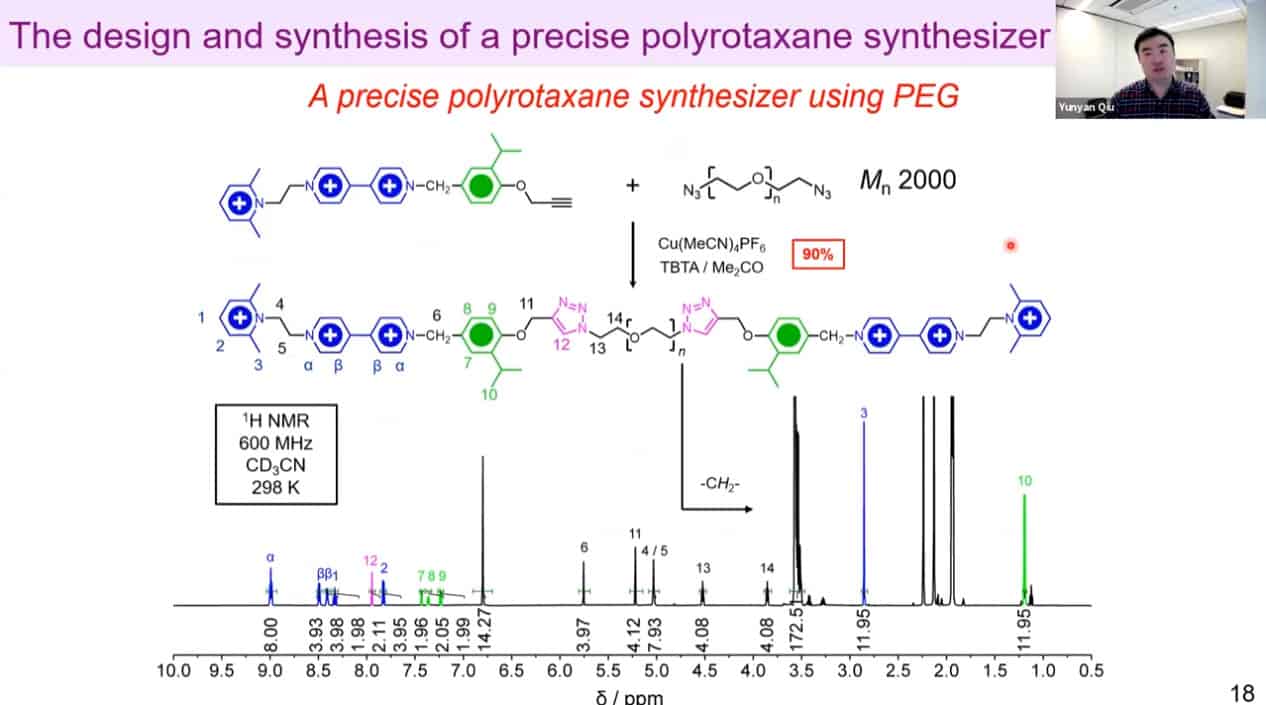
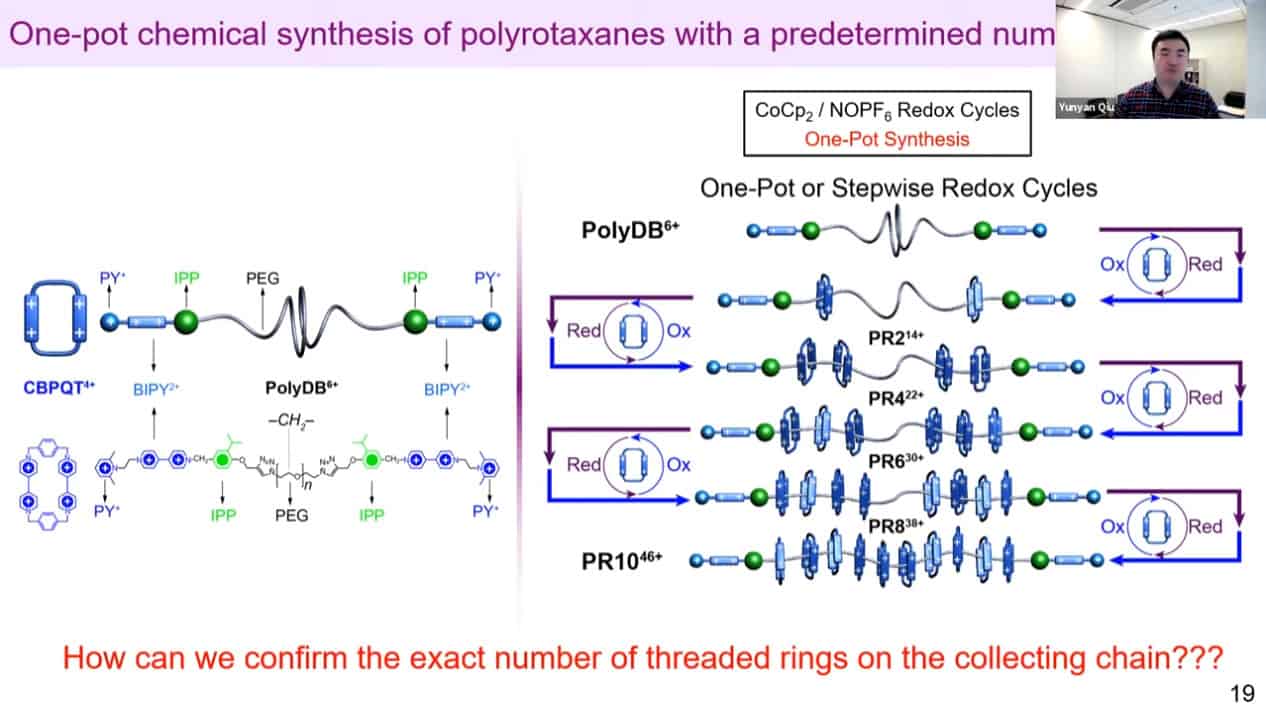
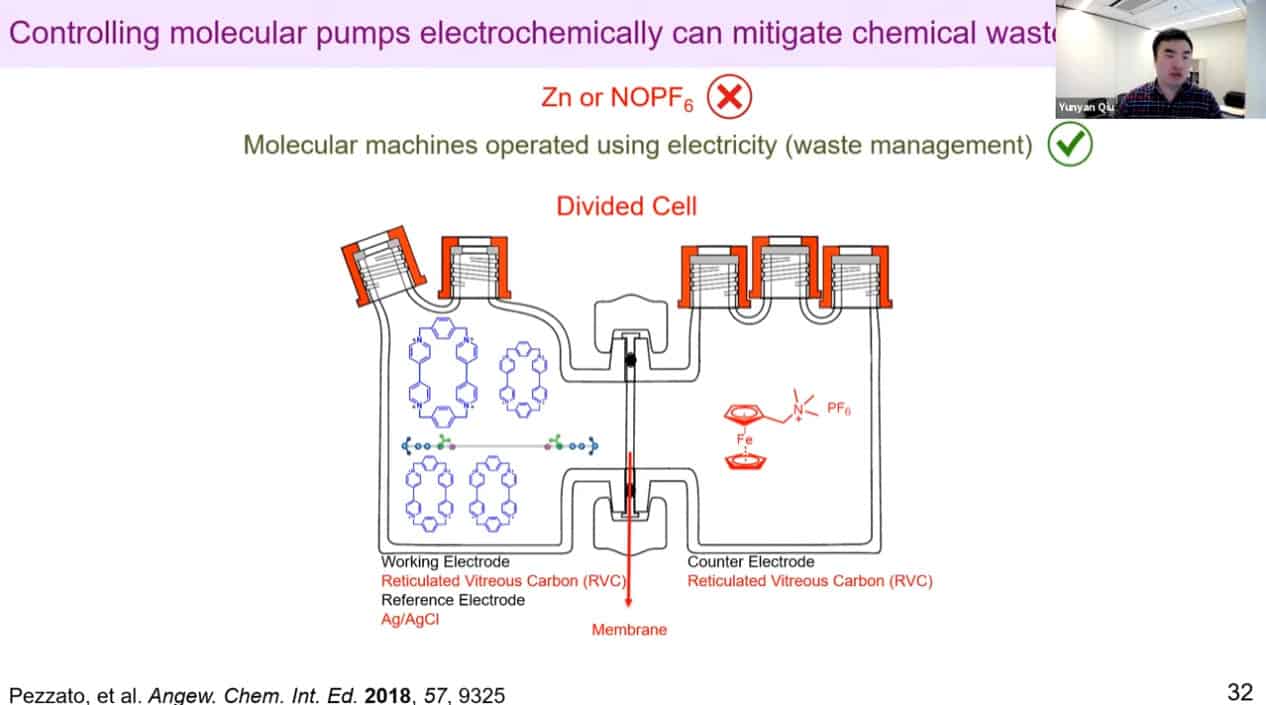
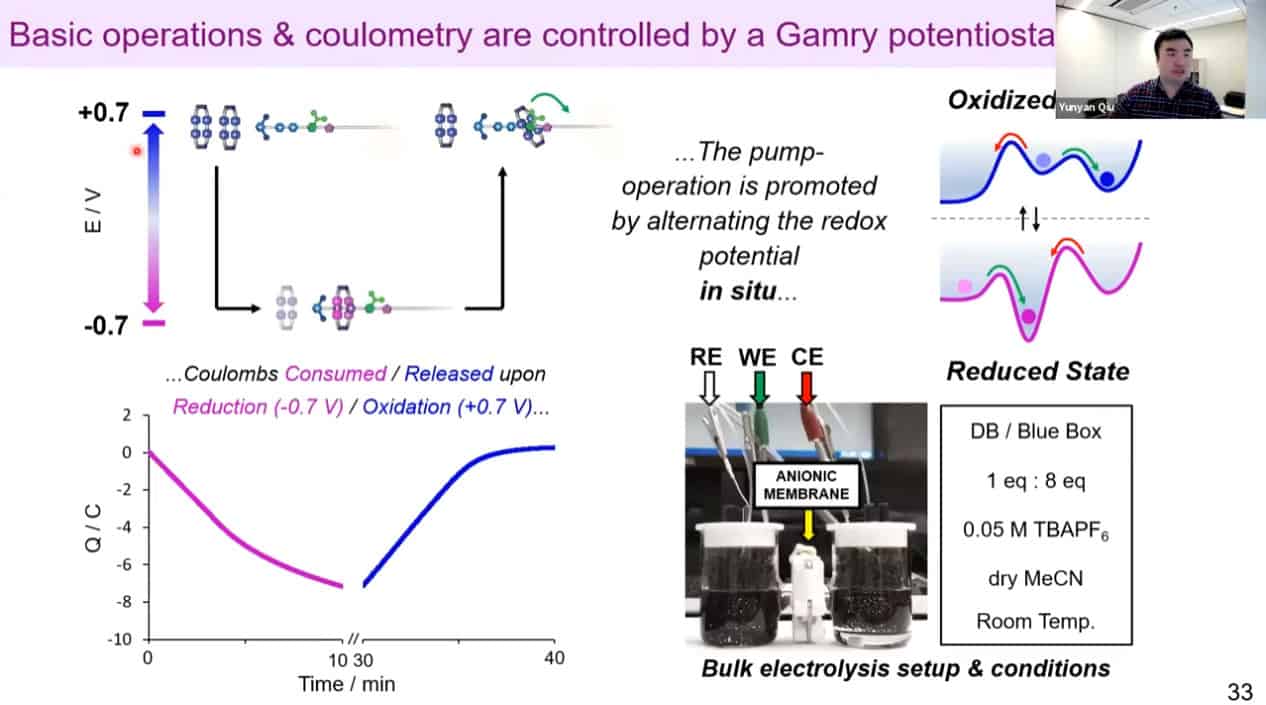
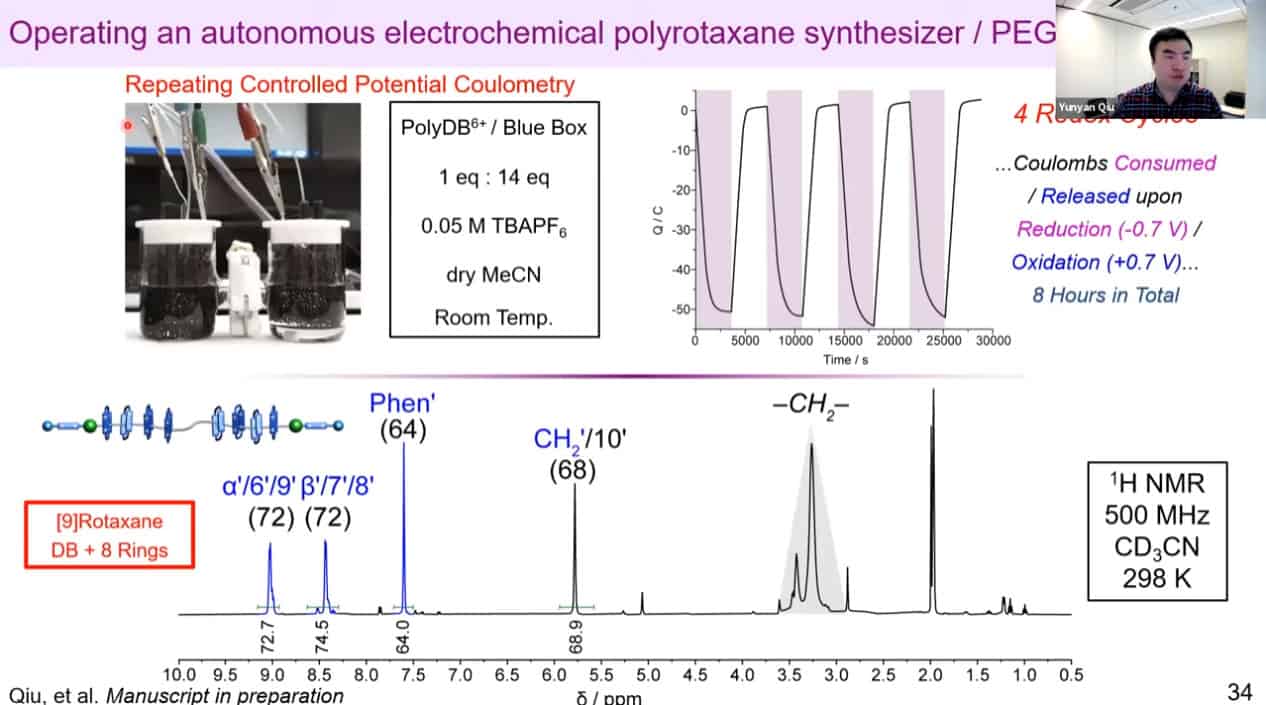
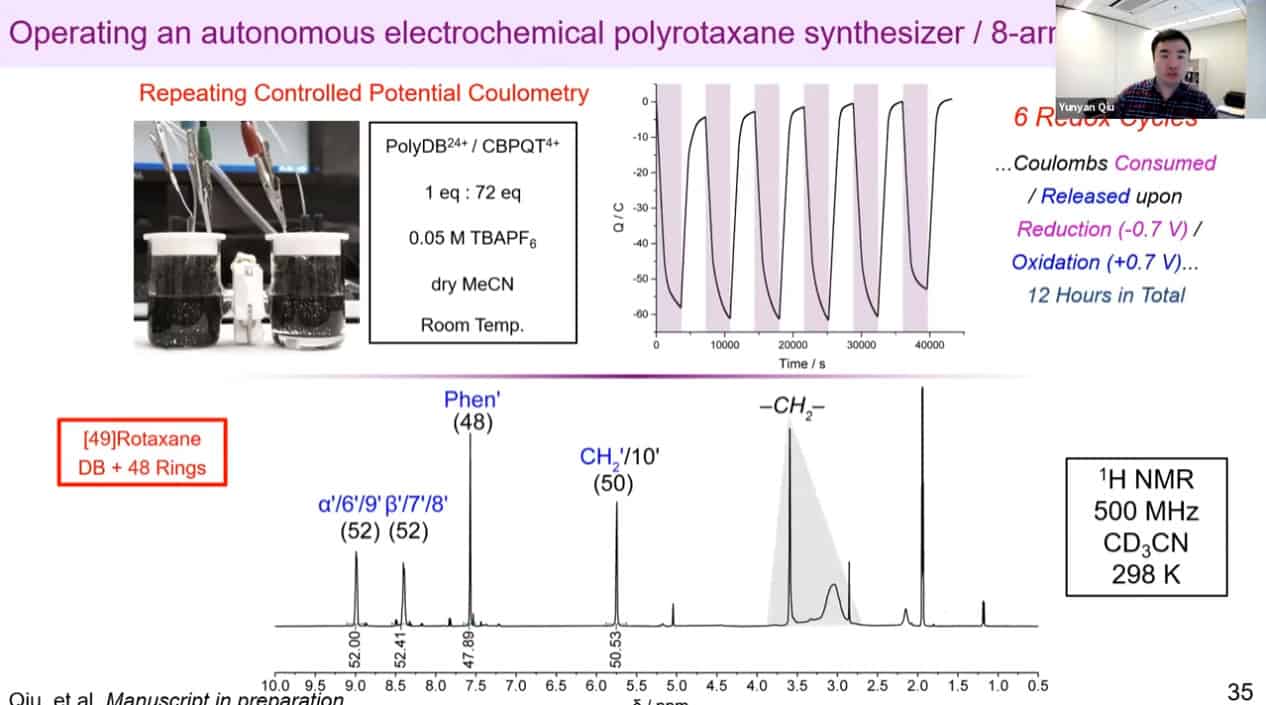
The molecular rotaxane pump can create precise polyrotaxane chains by doing redox cycling.
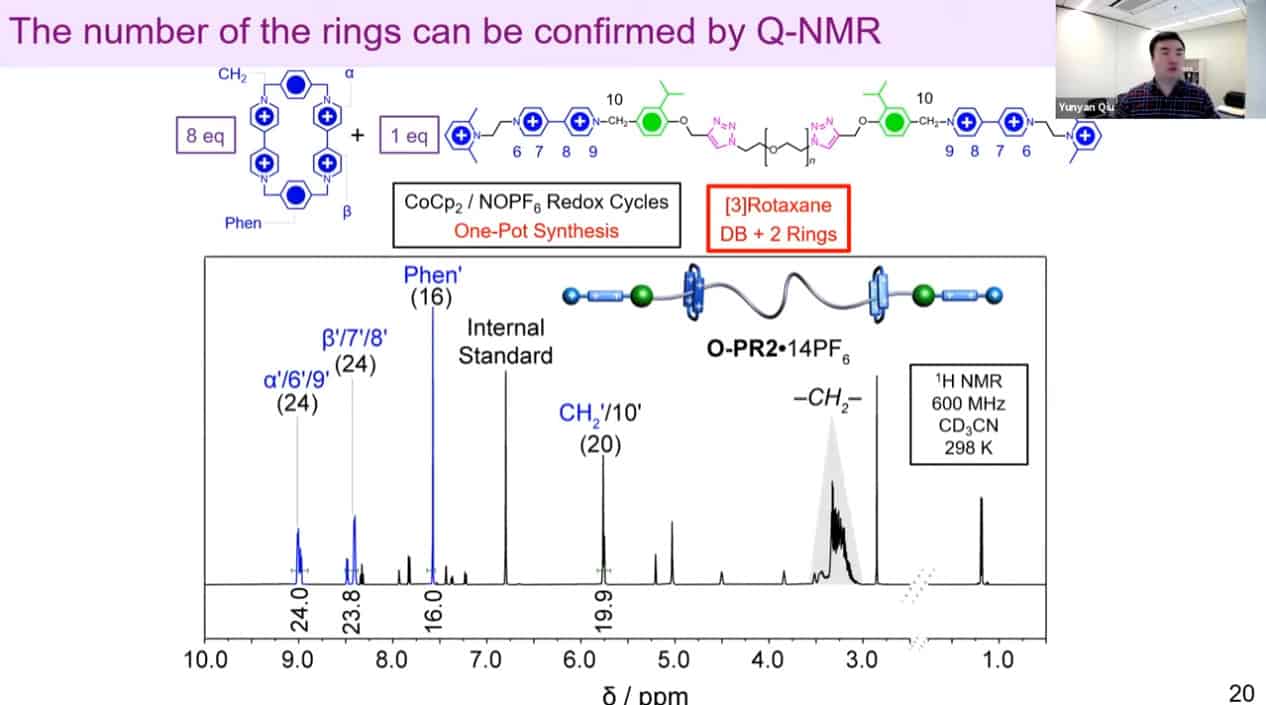
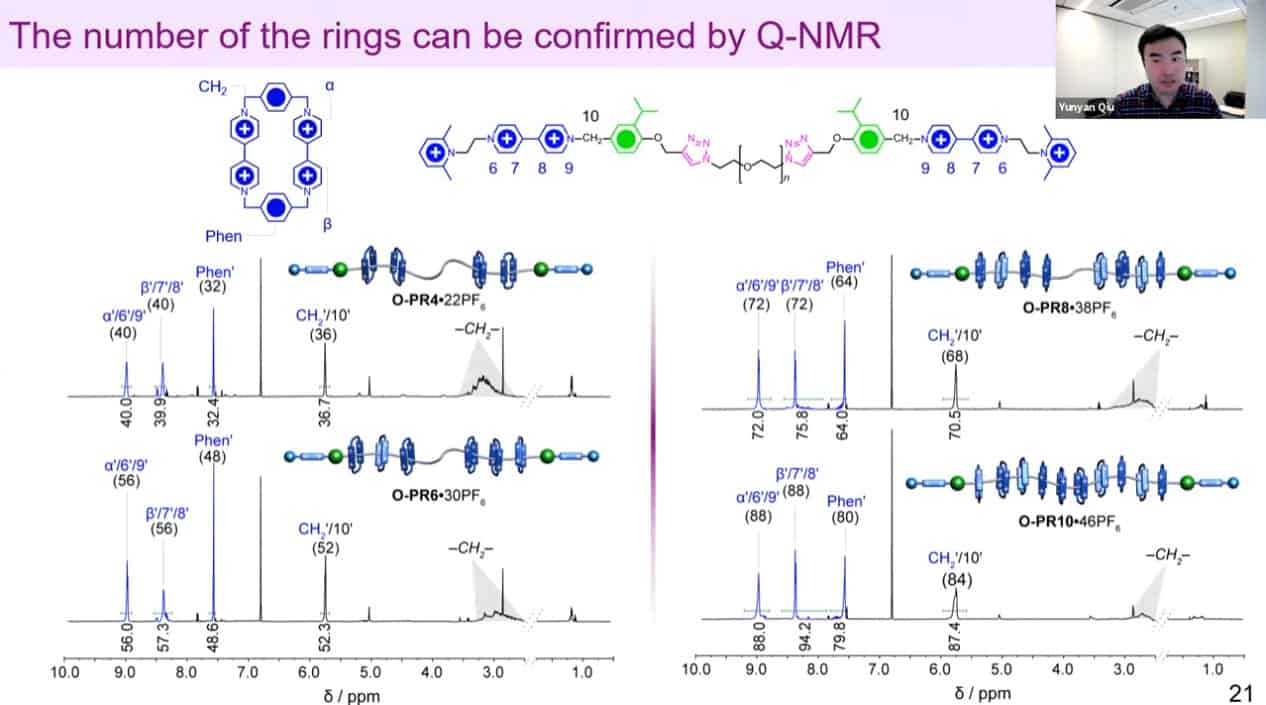
The number of threaded rings can be measured by quantitative NMR analysis to confirm the mechanism works.
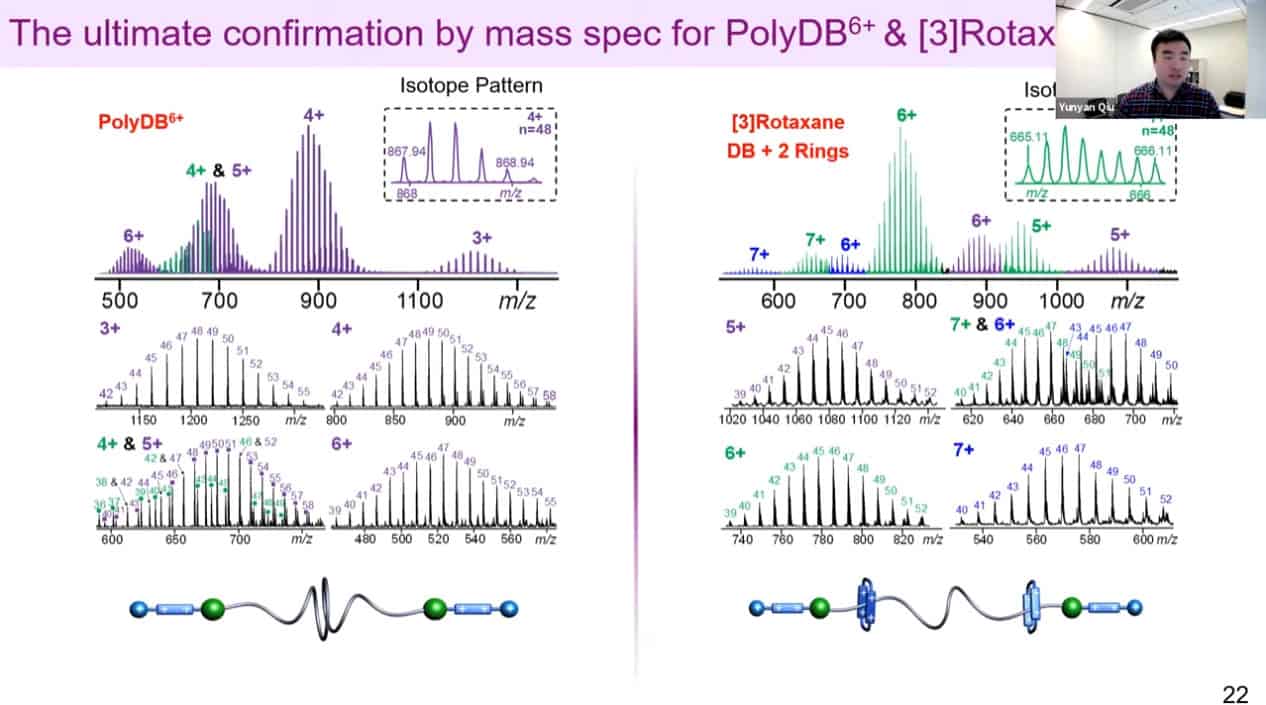
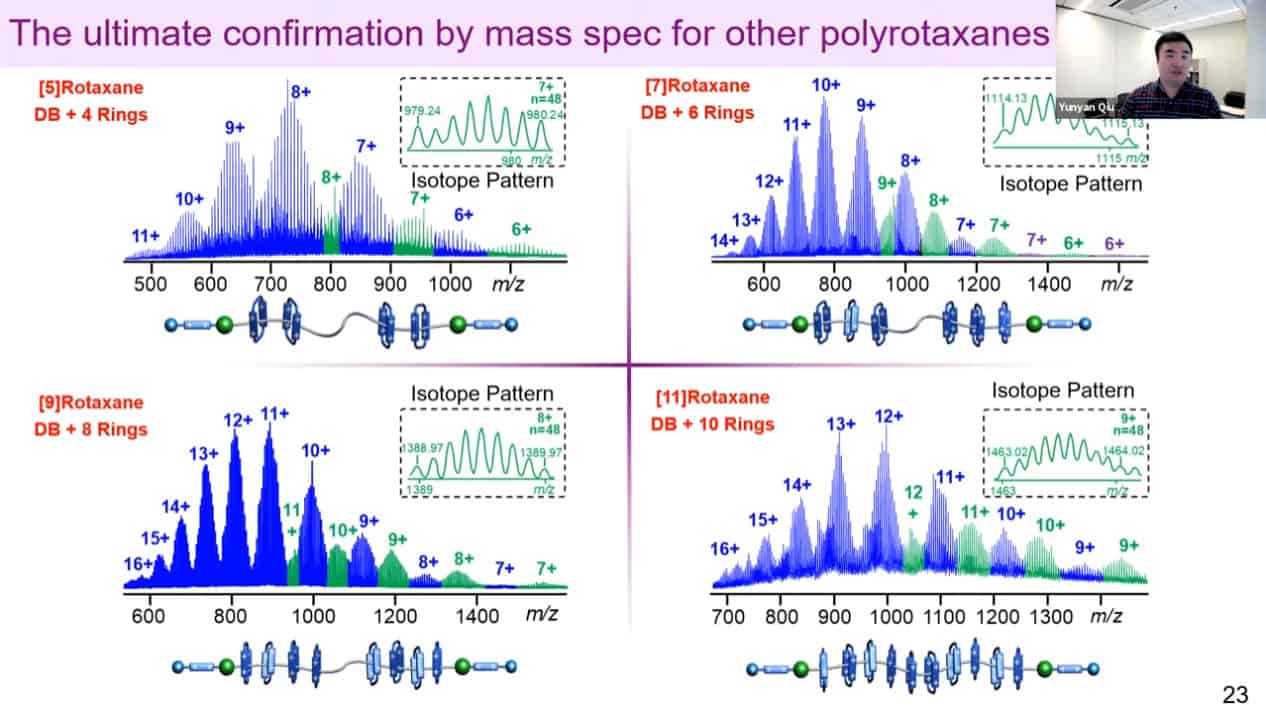
Mass spectrometry can be used to measure the number of rings on a polyrotaxane as well.
More analysis – hydrodynamic diameters.
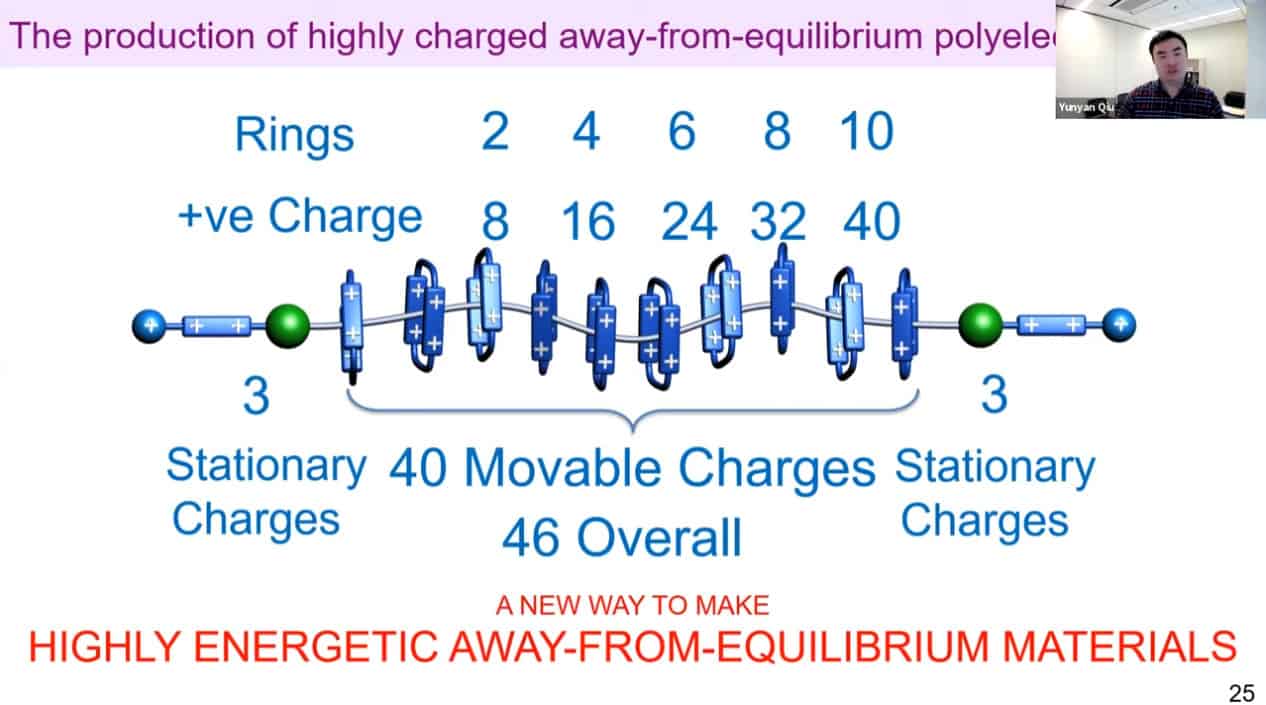
It is possible to produce highly energetic away-from-equilibrium materials by using artificial molecular pumps. Maybe there are applications for energetic materials such as battery storage.
Redox reactions for polyrotaxane construction can be controlled via electricity. By cycling electrical charge, it is possible to automate the polyrotaxane production process. It is becoming difficult to use mass spec to measure the rotaxanes due to the large size of these assemblies.
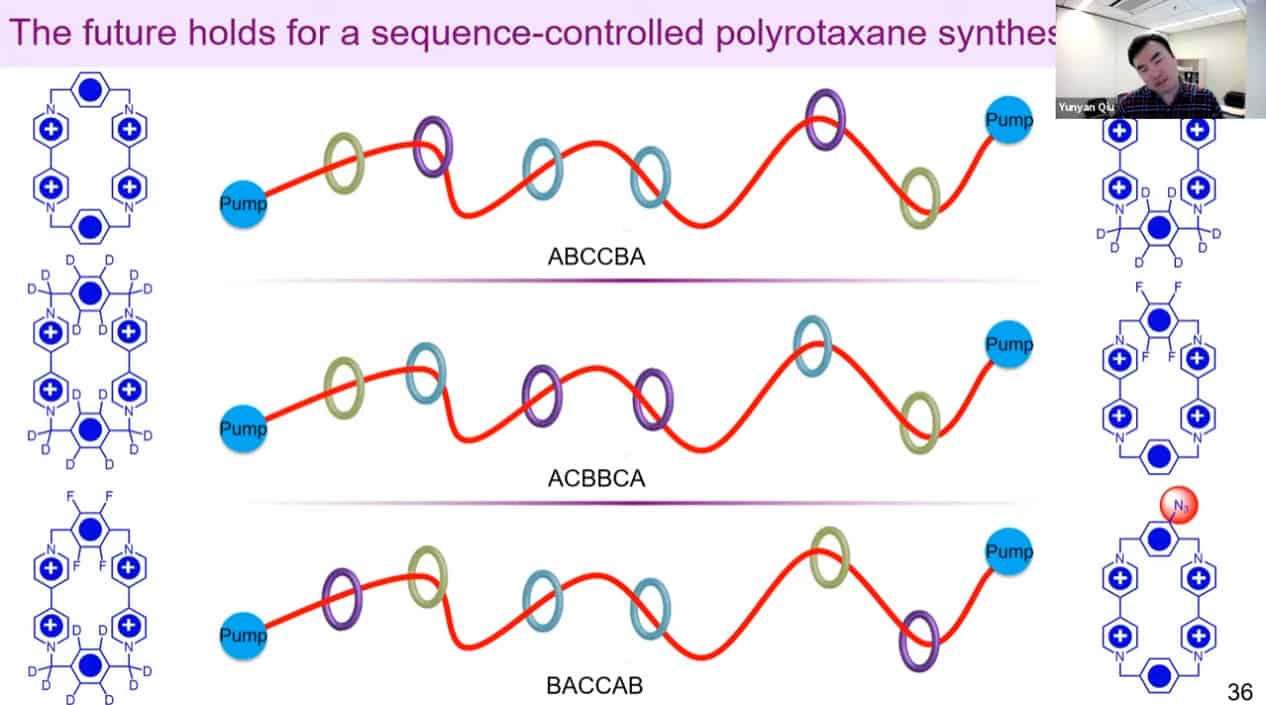
In the future, these molecular pump machines may be able to assemble long sequences of polymers.
Presentation: Active adsorption driven by molecular pumps
Liang Feng
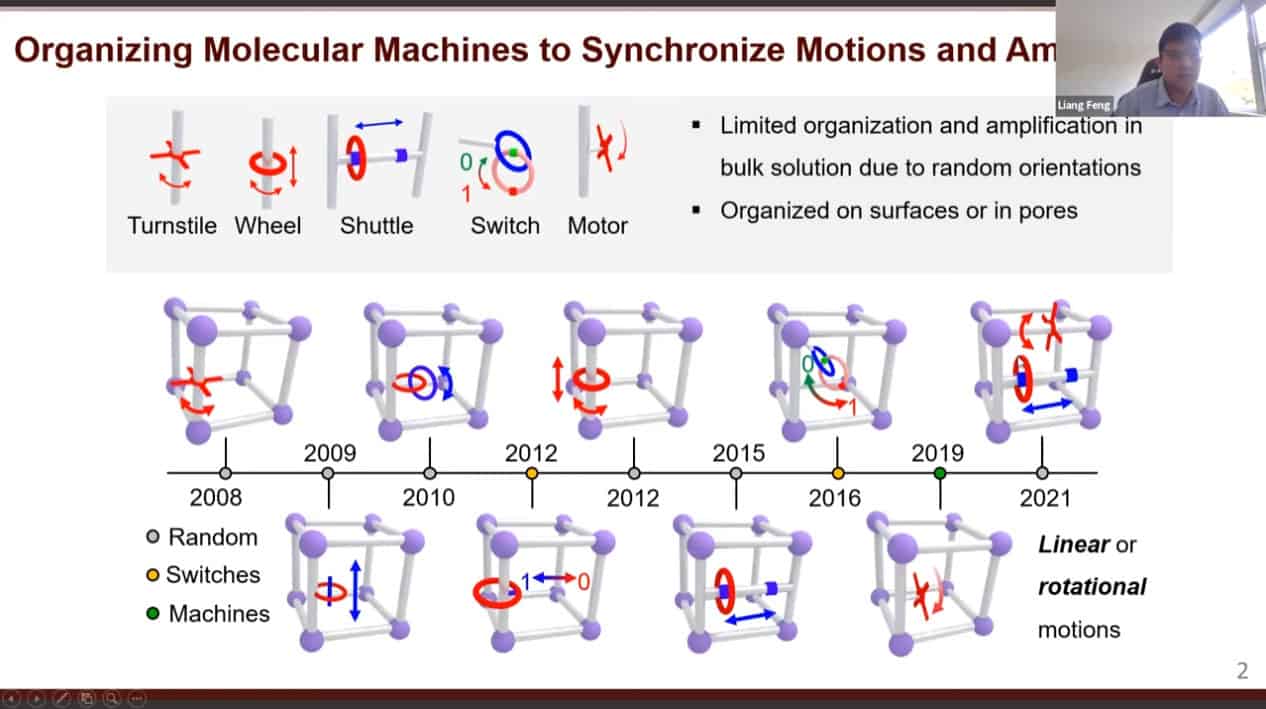
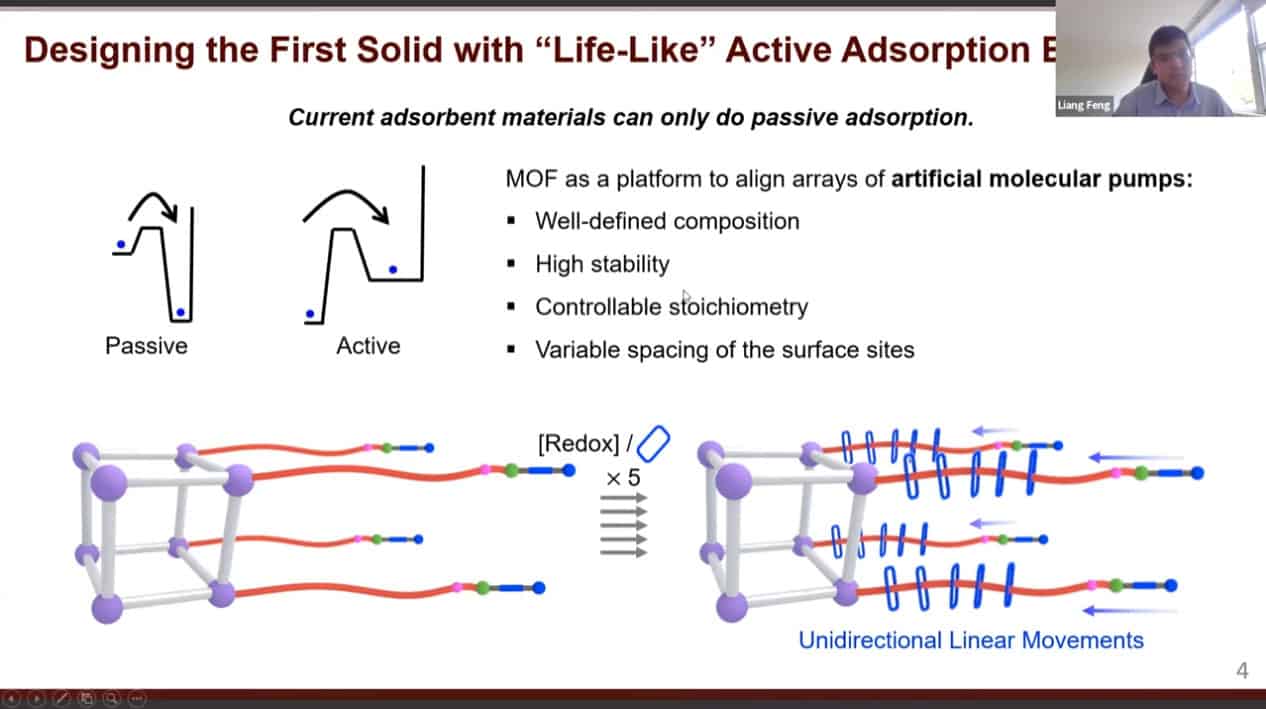
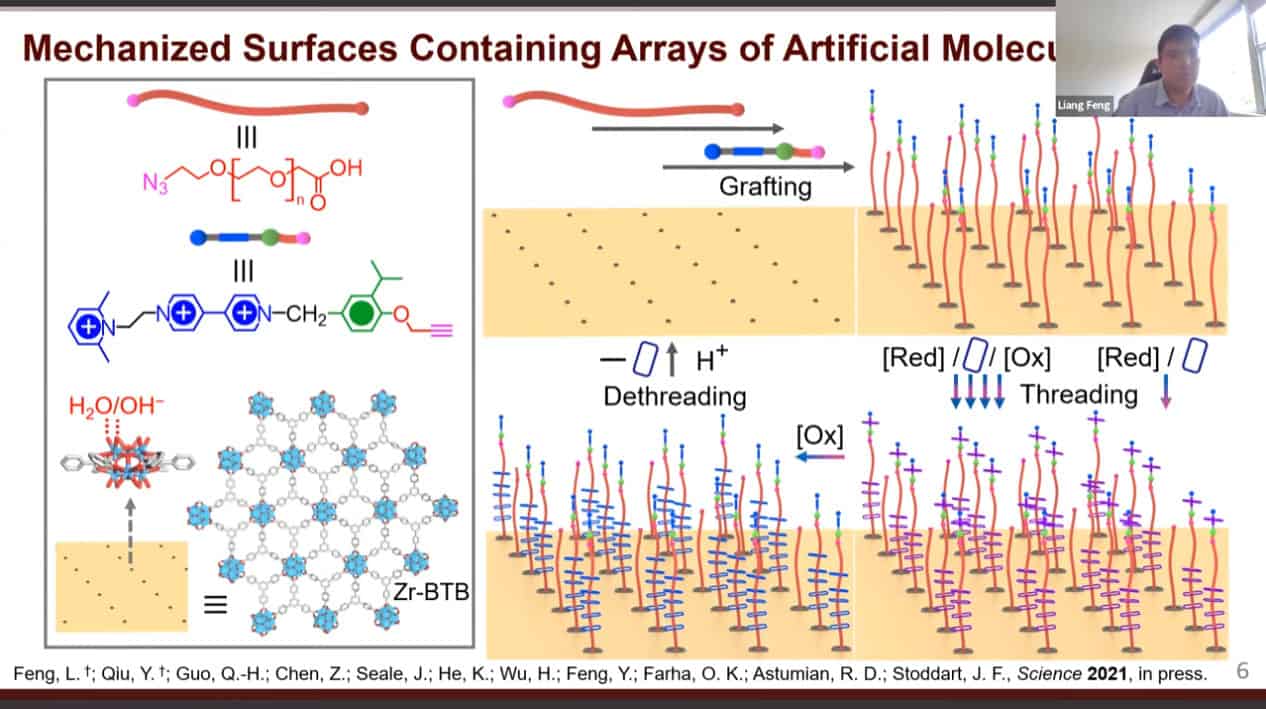
- There are a large variety of dynamic molecules that have been studied. However, when floating in solution they show very limited organization. Organizing these machines on solid materials can amplify their output. We can construct porous materials with very large pores then place molecular machines within them.
- Molecular pumps can generate polyrotaxanes in solution.
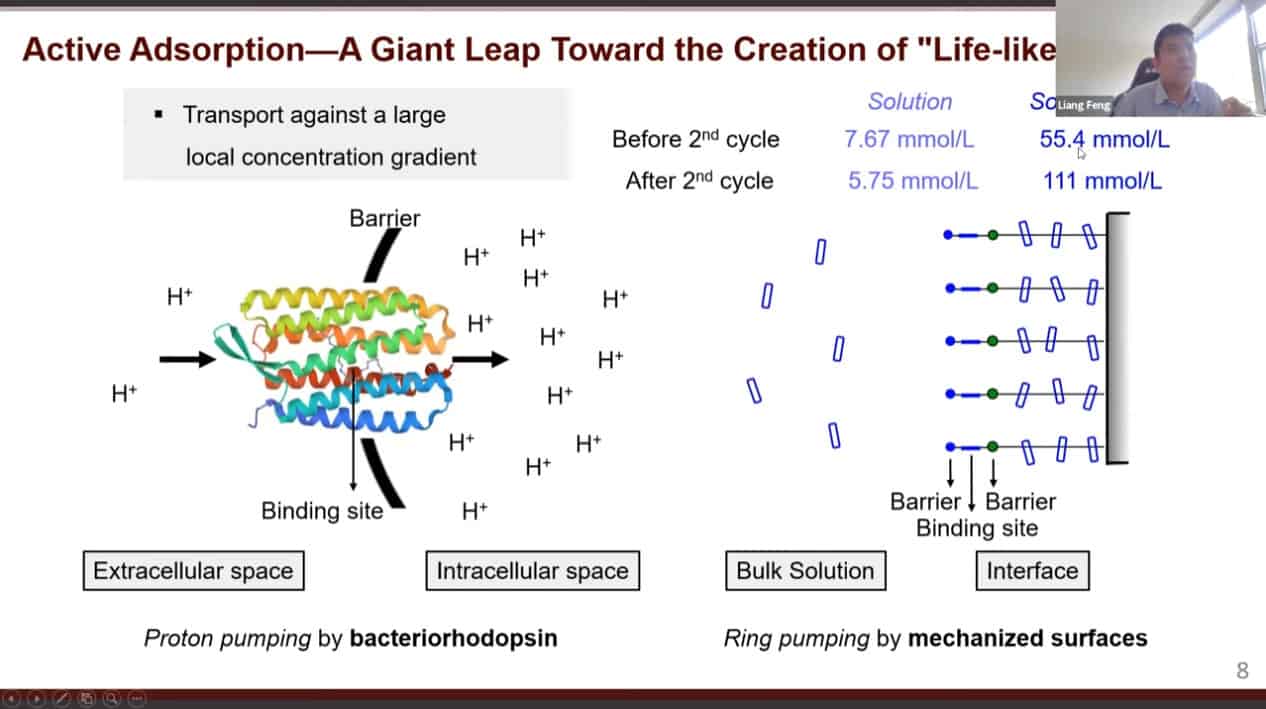
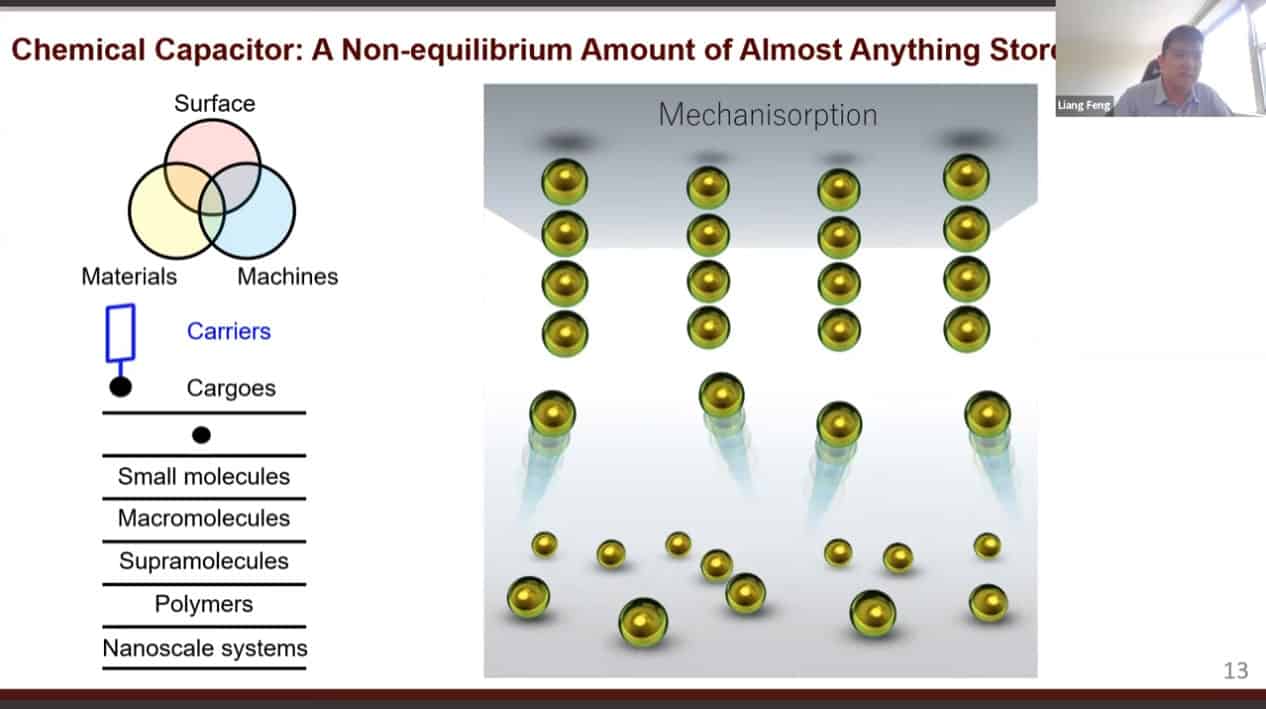
We can use molecular pumps and rotaxanes to create a material with active adsorption – able to move material from an area of low concentration to an area of high concentration.
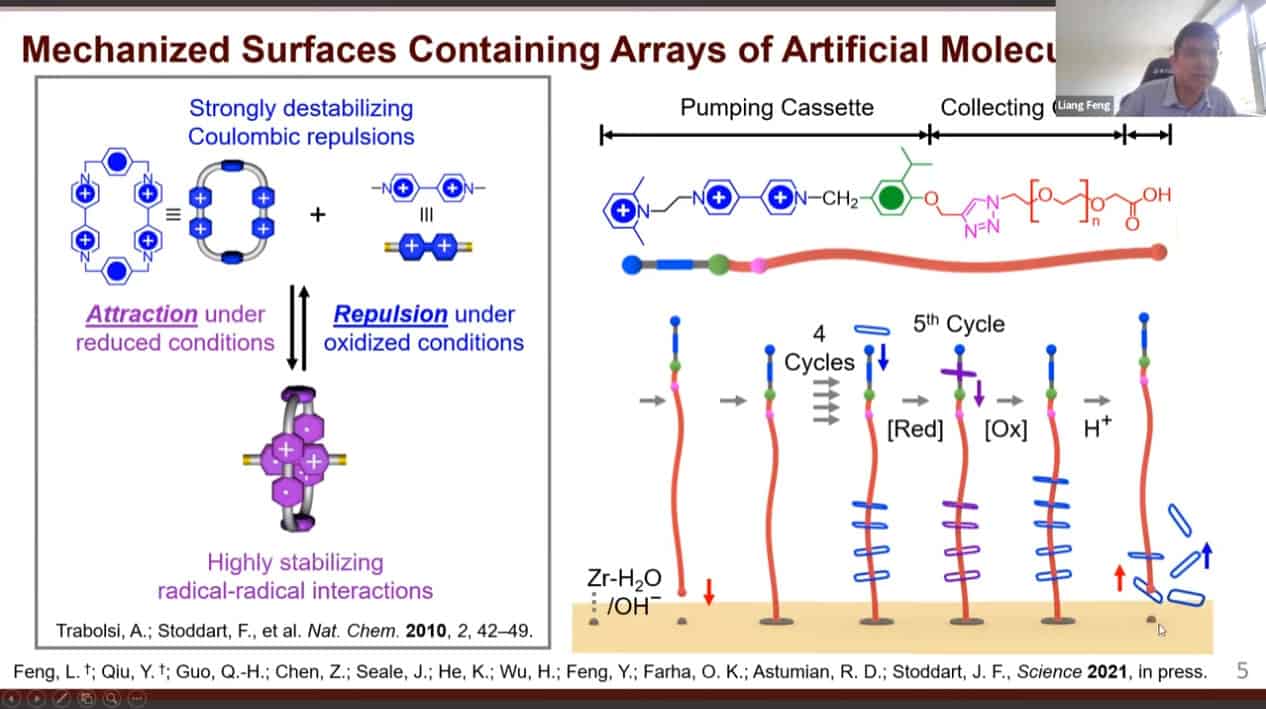
Cycling redox reactions will attract substrates to the pump cassette then move those substrates onto the collecting chain.
This mechanism is very similar to biological pumps which transport protons against a concentration gradient.
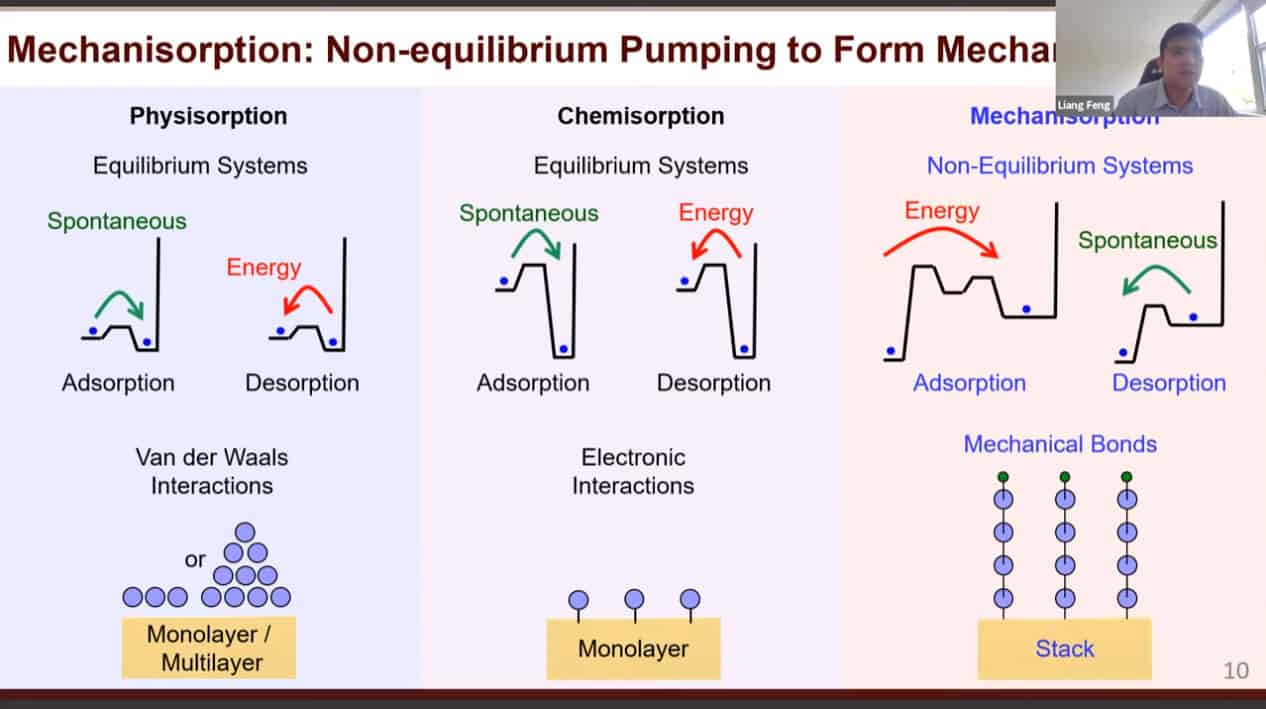
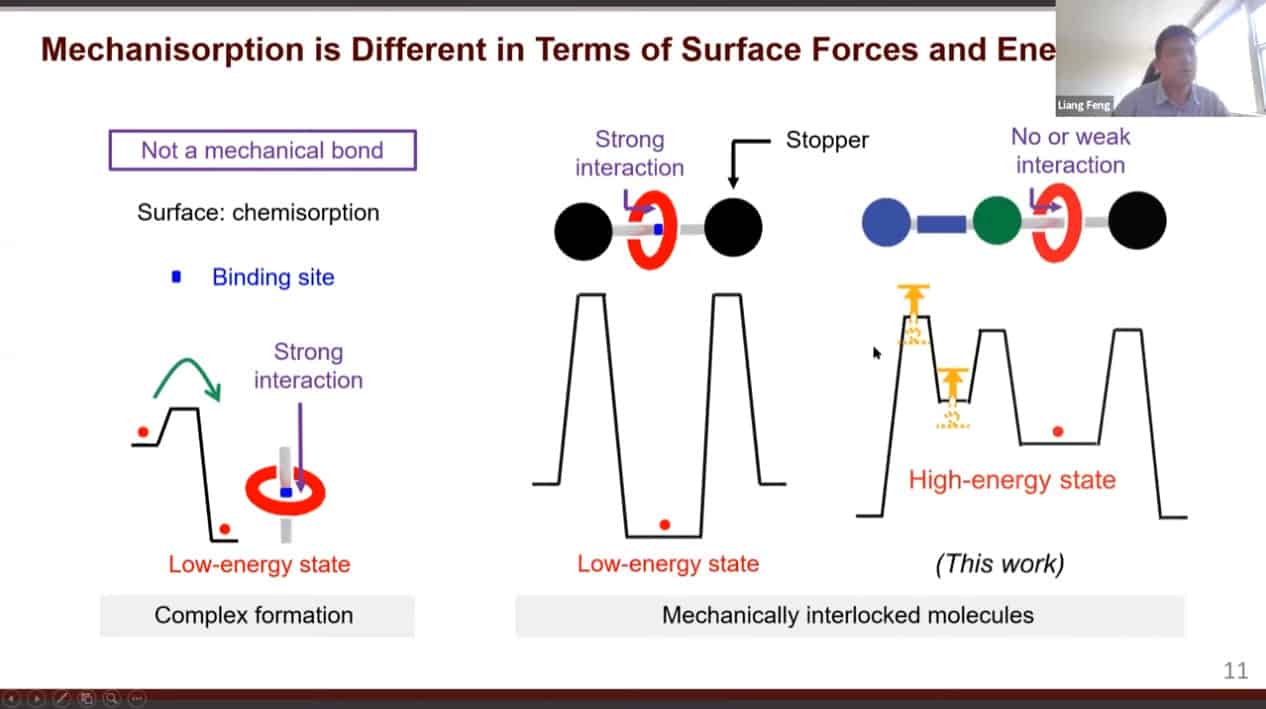
Physical and chemical adsorption requires energy for desorption. However, in mechanisorption the energy is required for adsorption while desorption is spontaneous. The bonds keeping things adsorbed are mechanical in nature.
It may be possible to control the sequences of synthetic polymers using this technology.
Seminar summary by Aaron King.