Our most recent posts (here and here) focused on increasing acceptance of the idea that the ultimate future of nanotechnology rests with high throughput atomically precise manufacturing. This one exemplifies the use of atomically precise elements from biotechnology and chemistry incorporated into non-atomically precise but increasingly sophisticated nanostructures to advance one application area—medicine. In this case the area of medical interest is the number one cause of death in the industrial world: cardiovascular disease. Foresight President Paul Melnyk forwards this link “Nanotechnology ‘could signal the future of medicine’, scientists claim“:
Microscopic drones which can seek out and repair sections of artery damage could signal the future of treatments for heart disease and strokes, scientists claim.
Successful tests of the nanodrones have been carried out in mice – and researchers hope to conduct the first human trials soon.
The tiny particles are 1,000 times smaller than the tip of a human hair, and are designed to latch on to atherosclerotic plaques – hard deposits made from accumulated fat, cholesterol and calcium that build up on the walls of arteries and are prone to rupture, producing dangerous clots.
Once they have attached, they release a drug derived from a natural protein which can repair damage in the body.
In the mice, scientists found that just five weeks of treatment resulted in significant repairs to artery damage while the plaques were shrunk and stabilised, making it less likely for fragments to break off and cause clots.…
This work is the fruit of a collaboration between the laboratory of Ira A. Tabas at Columbia University Health Sciences, which studies the cellular biology of advanced atherosclerotic plaque progression, and the laboratory of Omid Farokhzad at Brigham and Women’s Hospital at Harvard Medical School, which seeks to improve nanoparticle synthesis and formulation and its therapeutic efficacy. We have pointed to the work of Prof Farokhzad and his collaborators in the cancer therapy area several times (here, here, and here). A hat tip to Science Daily for reprinting this news release from Brigham and Women’s Hospital “Keeping Atherosclerosis in-check with Novel Targeted Inflammation-Resolving Nanomedicines“:
Nanometer-sized “drones” that deliver a special type of healing molecule to fat deposits in arteries could become a new way to prevent heart attacks caused by atherosclerosis, according to a study in pre-clinical models by scientists at Brigham and Women’s Hospital (BWH) and Columbia University Medical Center. These findings are published in the February 18th online issue of Science Translational Medicine [abstract, pdf reprint courtesy of Tabas Lab].
Although current treatments have reduced the number of deaths from atherosclerosis-related disease, atherosclerosis remains a dangerous health problem: Atherosclerosis of the coronary arteries is the #1 killer of women and men in the U.S., resulting in one out of every four deaths. In the study, targeted biodegradable nano ‘drones’ that delivered a special type of drug that promotes healing (‘resolution’) successfully restructured atherosclerotic plaques in mice to make them more stable. This remodeling of the plaque environment would be predicted in humans to block plaque rupture and thrombosis and thereby prevent heart attacks and strokes.
“This is the first example of a targeted nanoparticle technology that reduces atherosclerosis in an animal model,” said co-senior author Omid Farokhzad, MD, associate professor and director of the Laboratory of Nanomedicine and Biomaterials at BWH and Harvard Medical School (HMS). “Years of research and collaboration have culminated in our ability to use nanotechnology to resolve inflammation, remodel and stabilize plaques in a model of advanced atherosclerosis.”
In this study, targeted nanomedicines made from polymeric building blocks that are utilized in numerous FDA approved products to date, were nanoengineered to carry an anti-inflammatory drug payload in the form of a biomimetic peptide. Furthermore, this peptide was derived from one of the body’s own natural inflammatory-resolving proteins called Annexin A1. The way the nanomedicines were designed enabled this biological therapeutic to be released at the target site, the atherosclerotic plaque, in a controlled manner.
In mouse models with advanced atherosclerosis, researchers administered nanomedicines and relevant controls. Following five weeks of treatment with the nanomedicines, damage to the arteries was significantly repaired and plaque was stabilized.
Specifically, researchers observed a reduction of reactive oxygen species; increase in collagen, which strengthens the fibrous cap; and reduction of the plaque necrotic core, and these changes were not observed in comparison with the free peptide or empty nanoparticles.
“Many researchers are trying to develop drugs that prevent heart attacks by tamping down inflammation, but that approach has some downsides,” said co-senior author Ira Tabas, MD, Richard J. Stock professor of Medicine (Immunology) and professor of Pathology & Cell Biology at Columbia. “One is that atherosclerosis is a chronic disease, so drugs are taken for years, even decades. An anti-inflammatory drug that is distributed throughout the entire body will also impair the immune system’s ability to fight infection.” That might be acceptable for conditions that severely affect quality of life, like rheumatoid arthritis, but “using this approach to prevent a heart attack that may never happen may not be worth the risk.”
In addition, it’s not enough to deliver an anti-inflammatory drug to the plaques, said Columbia associate research scientist Gabrielle Fredman, PhD, one of the study’s lead co-authors. “Atherosclerosis is not only inflammation; there’s also damage to the arterial wall. If the damage isn’t repaired, you may not prevent heart attacks.”
The targeted nanomedicines used in this current study were engineered by researchers at BWH. Following preliminary proof-of-principle studies at Columbia University in models of inflammation, they were further tested in a clinically relevant disease model in mice and were shown to be capable of maneuvering through the blood circulation, and traversing leaky regions through to the inside of the plaques, as was demonstrated by fluorescence microscopy imaging of the plaque lesions.
Researchers note that in addition to their specific ‘sticky’ surfaces, their small sub-100 nanometer size is also a key property that facilitates the retention and accumulation of these nanoparticles within the plaques. These nanoparticles are 1000 times smaller than the tip of a single human-hair strand.
“These nanomedicines are developed using biodegradable polymers that can break-up over time in the body using the bodies natural mechanisms, and can be nanoengineered using scale-able chemistries and nanotechnologies, which ultimately can facilitate their rapid translation to the clinic,” said co-lead author Nazila Kamaly, PhD, instructor in the Laboratory of Nanomedicine and Biomaterials at BWH and HMS.
Researchers caution that although plaques in mice look a lot like human plaques, mice do not have heart attacks, so the real test of the nanoparticles will not come until they are tested in humans. “In this study, we’ve shown, for the first time, that a drug that promotes resolution of inflammation and repair is a viable option, when the drug is delivered directly to plaques via nanoparticles,” said Tabas. To be ready for testing in humans, the team plans to fine-tune the nanoparticles to optimize drug delivery and to package them with more potent resolution-inducing drugs. “We think that we can obtain even better delivery to plaques and improve healing more than with the current peptides,” , he said.
Farokhzad and colleagues have considerable expertise with bench-to-bedside translation of nanotechnologies for medical applications, and foundational work done in part by his team has led to the development and first in human testing of a targeted nanoparticle capable of controlling drug release for treatment of cancers, and the first in human testing of a targeted nanoparticle vaccine capable of orchestrating an immune response to facilitate smoking cessation and relapse prevention.
“The inflammation resolving targeted nanoparticles have shown exciting potential not only for the potential treatment of atherosclerosis as described here, but also other therapeutic areas including wound repair, for example, as described in the Feb. 9 online issue of Journal of Clinical Investigation, in addition to other applications currently underway with our collaborators,” Farokhzad said. “I’m optimistic that with additional animal validation we will also consider the human testing of the inflammation resolving targeted nanoparticles for a myriad of unmet medical needs—these are exciting times in medicine and the future of nanomedicine is incredibly bright.”
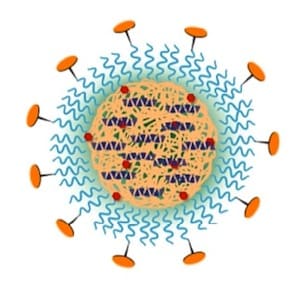
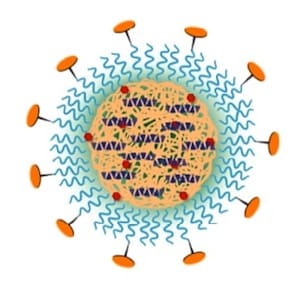
The very promising core-shell nanoparticle platform used here employs a hydrophobic interior comprising PLGA, a block copolymer of lactic and glycolic acids—both natural metabolic products—and diagnostic and therapeutic cargo molecules. Some PLGA molecules are linked to a fluorescent dye to track the disposition of the nanoparticles in animals. Encapsulated with the PLGA are molecules of a 25-amino acid peptide that acts to resolve inflammation through binding to important cell surface receptors. Some PLGA molecules are attached at one end to the hydrophilic polyethylene glycol (PEG), which forms the outer shell of the nanoparticle. Attachment of a peptide to the other end of PEG targets the nanoparticle to a collagen protein found in the vascular basement membrane that becomes exposed with vascular injury and inflammation.
Impressive evidence of the efficacy of these sophisticated nanoparticles is presented. Most of the discussion section at the end of the paper focuses on the complexity of the molecular and cellular mechanisms involved with vascular injury, especially in very advanced lesions. It may very well be that nanoparticles of the type presented here will prove more than adequate to optimize the desired clinical response. Or it may be that enough different molecules and mechanisms are important that it may be necessary to employ swarms of nanorobots capable of digital computation to provide the optimal response for each patient.
—James Lewis, PhD